Post-Transplant Cyclophosphamide Allows Allogeneic Hematopoietic Stem-Cell Transplantation Across Donor Types for Nonmalignant Hematologic Diseases
DOI:
https://doi.org/10.14740/jh2184Keywords:
Aplastic anemia, Diamond-Blackfan anemia, Allogeneic stem-cell transplantation, Conditioning chemotherapy, Graft-versus-host diseaseAbstract
Background: The aim of the study was to compare post-transplant cyclophosphamide (PTCY)-based regimens with historical regimens using calcineurin inhibitor and methotrexate (CNI-MTX) for allogeneic hematopoietic stem-cell transplant (HCT) in nonmalignant hematologic disorders.
Methods: We conducted a single-center, retrospective review of patients with acquired severe aplastic anemia (N = 18) or Diamond-Blackfan anemia (N = 1) who underwent allogeneic HCT from 2011 to 2024. Patients received graft-versus-host disease (GVHD) prophylaxis with either CNI-MTX or PTCY-mycophenolate mofetil-tacrolimus. Primary endpoints were overall survival (OS) and disease-free survival (DFS) without graft failure at 1 year after transplantation.
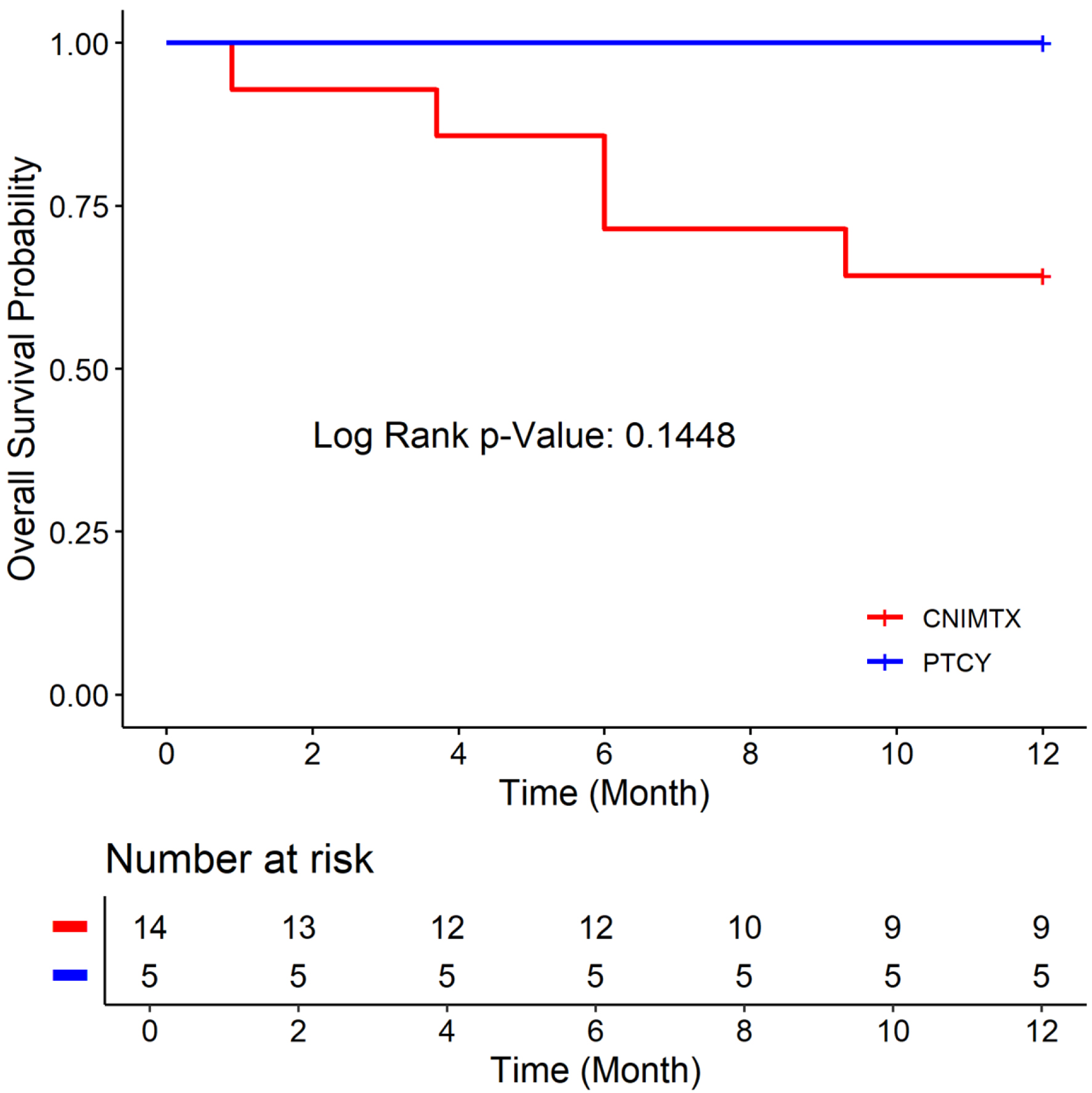
Results: In the CNI-MTX cohort (N = 14) with severe aplastic anemia, 11 patients received fludarabine-cyclophosphamide-thymoglobulin (ATG)-total body irradiation (TBI), while three received cyclophosphamide-ATG allogeneic HCT. Donors were matched-unrelated (N = 7), matched-related (N = 6), or mismatched-unrelated (N = 1). Graft sources included bone marrow (N = 12) or peripheral blood stem cells (N = 2). One patient developed grade 3 skin acute GVHD, and none had chronic GVHD. There was primary graft failure (N = 6), stable mixed T-cell chimerism (N = 4), and 100% donor chimerism (N = 4). Four patients with primary graft failure underwent salvage second transplants at a median of 103 days (35–322) after the first transplant. Five patients with primary graft failure died at a median of 6 months (0.89–9.3) from the first transplant. The PTCY cohort (N = 5) included four patients with severe aplastic anemia and one with Diamond-Blackfan anemia. All underwent fludarabine-cyclophosphamide-ATG-TBI allogeneic HCT. Donors were matched-related (N = 1), matched-unrelated (N = 2), syngeneic (N = 1), or haploidentical (N = 1). Graft source was peripheral blood stem cells (N = 3) for matched-related, matched-unrelated, and syngeneic transplants, and bone marrow (N = 2) for haploidentical and matched-unrelated donor transplants. Donor chimerism was 100% (N = 3) and mixed chimerism (N = 2). All patients became transfusion-independent, and none developed GVHD or graft failure. The 1-year OS rate was 64.29% vs. 100%, the 1-year DFS rate was 57.14% vs. 100%, and the 1-year GVHD-free, graft failure-free survival (GRFS) was 50% vs.100% for the CNI-MTX and PTCY cohorts, respectively. Despite a trend toward better OS, DFS, and GRFS for PTCY, the OS, DFS, and GRFS time distributions were not statistically significantly different (P = 0.1448, 0.0919, and 0.0627, respectively).
Conclusion: Allogeneic HCT with uniform conditioning of fludarabine-cyclophosphamide-ATG-TBI with PTCY GVHD prophylaxis is effective for adults with severe aplastic anemia or Diamond-Blackfan anemia across donor types (matched-related, syngeneic, matched-unrelated, haploidentical) and should be prospectively compared with historical regimens using CNI-MTX GVHD prophylaxis.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








