Cryopreservation and Thawing of Ex Vivo Expanded Cord Blood Hematopoietic Stem Cells
DOI:
https://doi.org/10.14740/jh2167Keywords:
Cryopreservation, Thawing, Hematopoietic stem cells, Stem cell expansion, Stem cell agonist cocktail, Cell deathAbstract
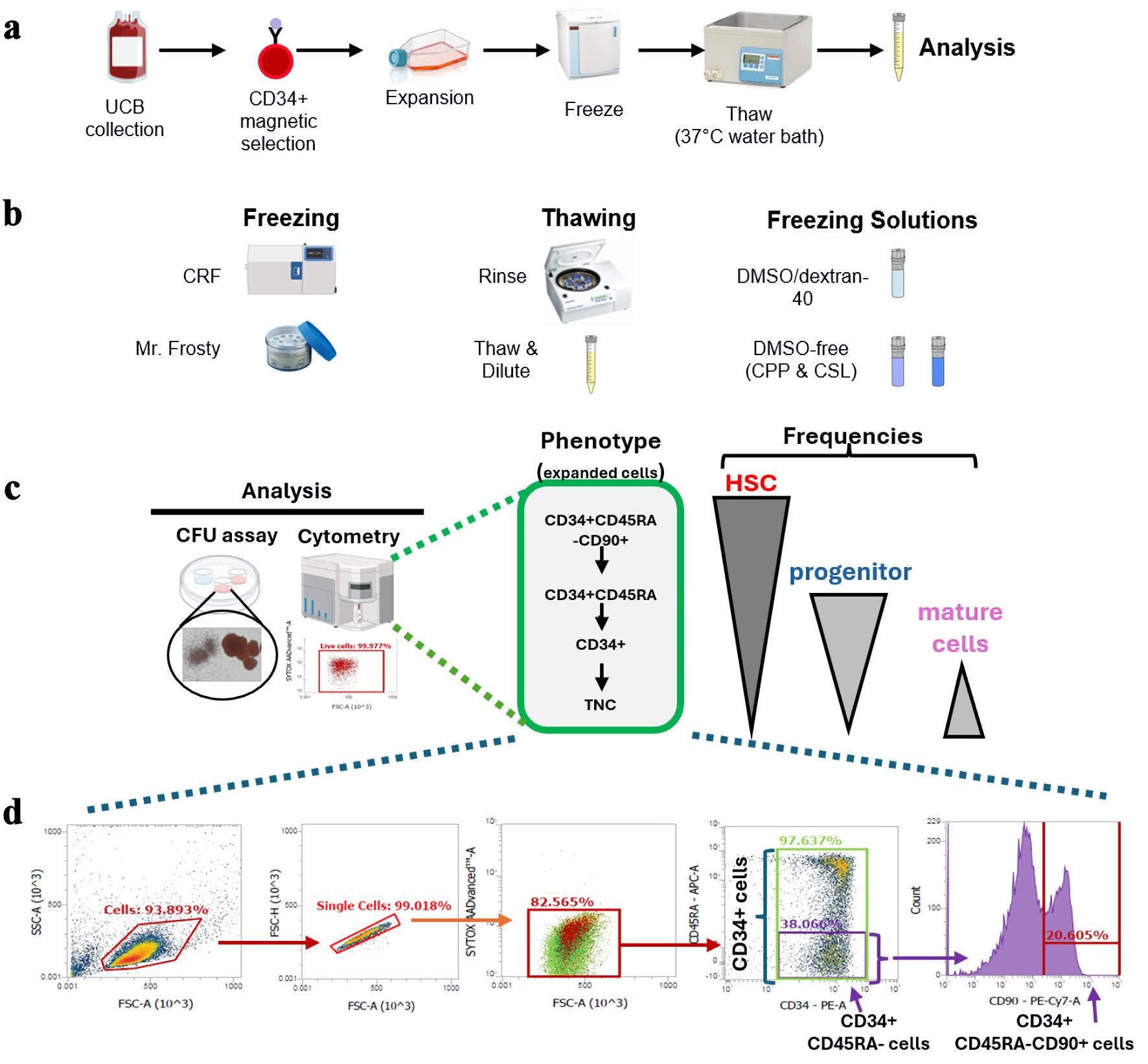
Background: Umbilical cord blood (CB) is an invaluable source of hematopoietic stem and progenitor cells (HSPCs). Its use in stem cell transplantation is however constrained by the insufficient cell dose present in each unit. Recent development in ex vivo HSPC expansion technologies addresses this issue and encourages the use of the best matched CB unit. In this study, we sought to develop a cryopreservation and thawing protocol for ex vivo expanded HSPC.
Methods: CB CD34+ HSPC-enriched cells were expanded in serum-free medium supplemented with a previously optimized mix of chromatin-modifiers and early acting cytokines for 7-days. CB HSPC were then harvested and prepared for cryopreservation. Thawed CB samples were then analyzed by flow cytometry to measure cell viability and recovery of HSPC-enriched fractions, while graft potency was measured using the colony-forming unit (CFU) assay.
Results: First, we compared two widely used means of freezing; a passive isopropyl alcohol-based freezing container vs. a controlled-rate freezer (CRF). Both methods exhibited comparable recovery of viable cell numbers, including the HSC-enriched CD34+CD45RA–CD90+ fraction, and similar potency measured using the CFU assay. Next, we compared two thawing methods frequently used in clinical settings. The “thaw and dilute” method slightly improved the recovery of total nucleated cells (TNC) and HSPC fractions over the “rinse” method, though potency was comparable between both thaw methods. Next, we investigated the impact of three different commercial freezing solutions on product recovery. Dimethyl sulfoxide (DMSO)/dextran-40 and CryoProtectPure-STEM (CPP) provided superior recovery of HSPC-fractions and potency when compared to CryoScarless (CSL).
Conclusions: Taken together, this study provides insights into alternative, less harmful options for the freezing and thawing of ex vivo expanded HSPCs.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








