Combined Impact of Prior Polatuzumab Vedotin Plus Bendamustine and Rituximab Therapy and Myeloablative Conditioning on Early Post-Transplant BK Virus-Associated Hemorrhagic Cystitis
DOI:
https://doi.org/10.14740/jh2010Keywords:
BK virus, Hemorrhagic cystitis, Allogeneic transplantation, Pola-BR, Polatuzumab, Relapsed/refractory diffuse large B-cell lymphomaAbstract
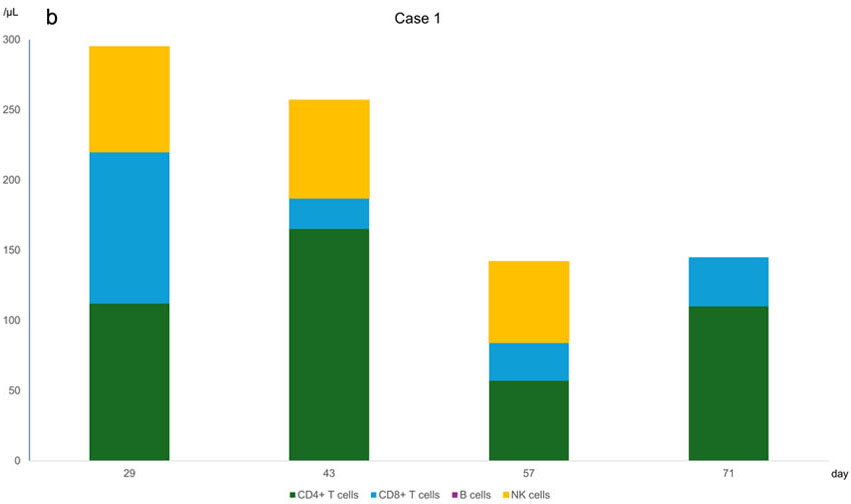
Relapsed/refractory diffuse large B-cell lymphomas (R/R DLBCLs) have an extremely poor prognosis, with no established salvage chemotherapy currently available. Polatuzumab, rituximab, and bendamustine combination therapy (Pola-BR) has been approved as a new therapeutic option for R/R DLBCL. Recently, chimeric antigen receptor T-cell therapy and bispecific antibodies have induced long-term remission in many patients with R/R DLBCL. However, allogeneic transplantation remains potentially curative for patients unresponsive to the abovementioned treatments. While allogeneic transplantation can also cause various adverse events, hemorrhagic cystitis is a particularly severe complication that requires effective prevention strategies. Here, we report two cases of severe BK virus-associated hemorrhagic cystitis (BKV-HC) that developed after successive cord blood transplantation with myeloablative conditioning and Pola-BR treatment for early-relapsed DLBCL. Both patients received Pola-BR after undergoing multiple salvage therapies and developed early-onset BKV-HC post-transplant, demonstrating the effects of Pola-BR and myeloablative conditioning. We analyzed the shared characteristics between these two cases to distinguish between the factors that trigger the onset of BKV-HC and those that contribute to its severity. Based on the differences in the clinical course between the two cases, we propose prevention strategies for BKV-HC and identify treatment strategies for Pola-BR in patients with R/R DLBCL undergoing allogeneic transplantation.
Published
Issue
Section
License
Copyright (c) 2025 The authors

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.








