Engraftment Outcome of CRISPR/Cas9-Edited Hematopoietic Stem Cells for Genetic Diseases: A Systematic Review and Meta-Analysis of Preclinical Evidence
DOI:
https://doi.org/10.14740/jh2190Keywords:
Hematopoietic stem cell, Gene editing, Transplantation, Mice model, Engraftment efficiency, Peripheral blood, Bone marrow, SpleenAbstract
Background: CRISPR-Cas9 (clustered regularly interspaced short palindromic repeats and CRISPR-associated protein 9)-based gene editing represents a promising frontier for treating monogenic hematologic disorders. Several preclinical studies have demonstrated the transplantation efficiency of CRISPR-Cas9-mediated gene editing in hematopoietic stem and progenitor cells (HSPCs) using various animal models. Nonetheless, these studies have employed diverse gene-editing strategies, utilizing HSPCs from different origins and transplanting them into distinct mouse strains. The present study aimed to determine the optimum conditions for efficient engraftment of genetically modified HSPCs across various organs, thereby facilitating the translation of preclinical research into clinical applications.
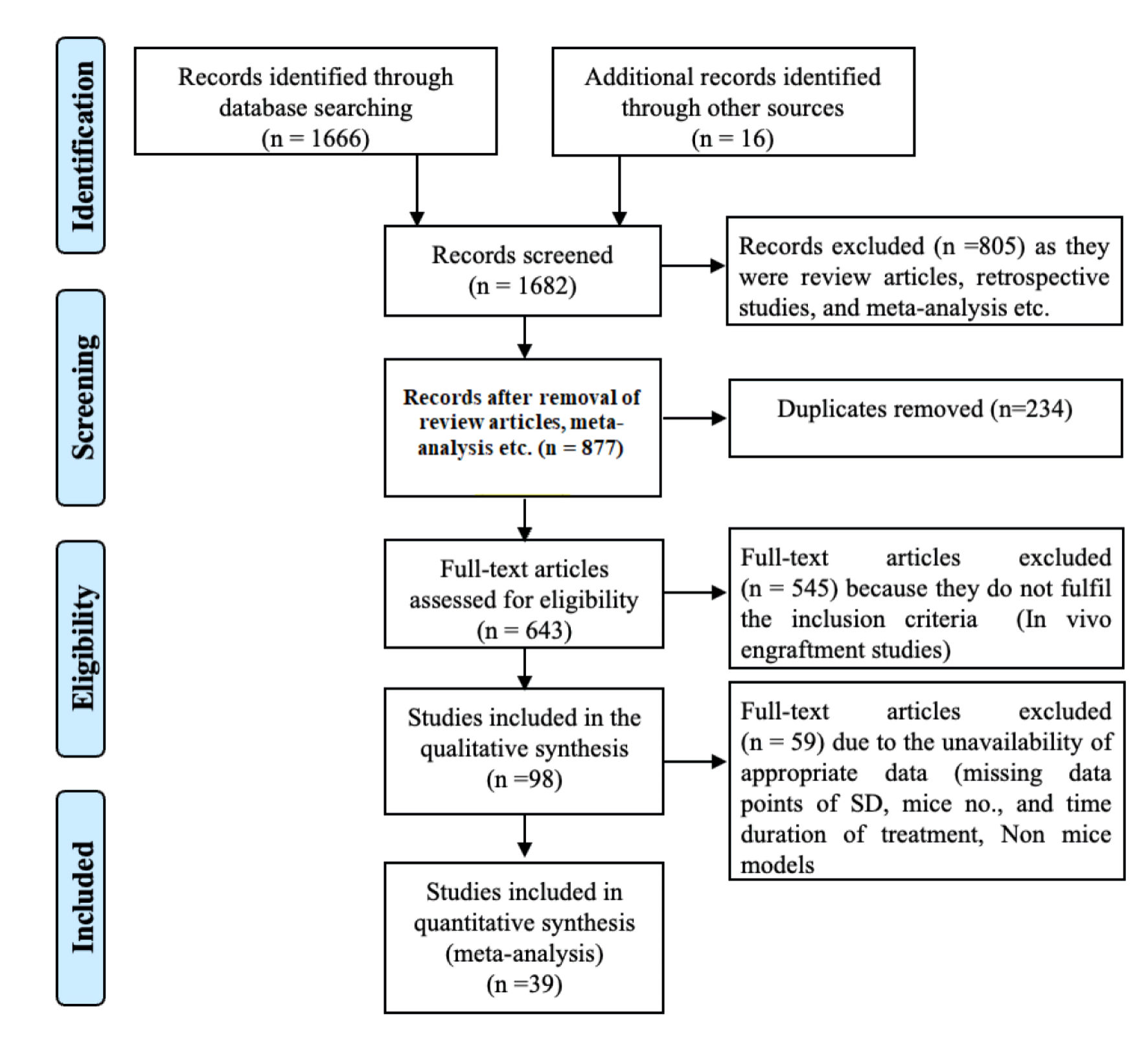
Methods: We conducted a comprehensive literature search using PubMed Medline, Web of Science, and Google Scholar for relevant articles published from 2014 to 2025 that evaluated the engraftment potential of CRISPR-Cas9 HSPCs in genetic disease models. A total of 39 studies met the inclusion criteria and were included in a meta-analysis using Jamovi software.
Results: The study revealed a significantly reduced engraftment of gene-edited cells in the bone marrow, spleen, and peripheral blood in the pooled analysis. Subgroup analyses revealed that knockout cells exhibited diminished engraftment, whereas knock-in cells demonstrated engraftment levels comparable to those of their non-edited counterparts. No evidence of publication bias or substantial heterogeneity in the study design or outcomes was detected.
Conclusions: Identifying the optimal parameters for gene editing to enhance engraftment efficiency may provide crucial insights for designing future clinical trials and advancing the therapeutic application of CRISPR-Cas9 edited HSPCs.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








