Adriamycin, Vinblastine and Dacarbazine With Immunotherapy Achieves Complete Metabolic Response in a Patient With Classical Hodgkin Lymphoma and Dyskeratosis Congenita
DOI:
https://doi.org/10.14740/jh2160Keywords:
Dyskeratosis congenita, Telomeropathy, Classical Hodgkin lymphoma, Brentuximab vedotin, BV-AVD, Chemotherapy toxicity, Radiation omissionAbstract
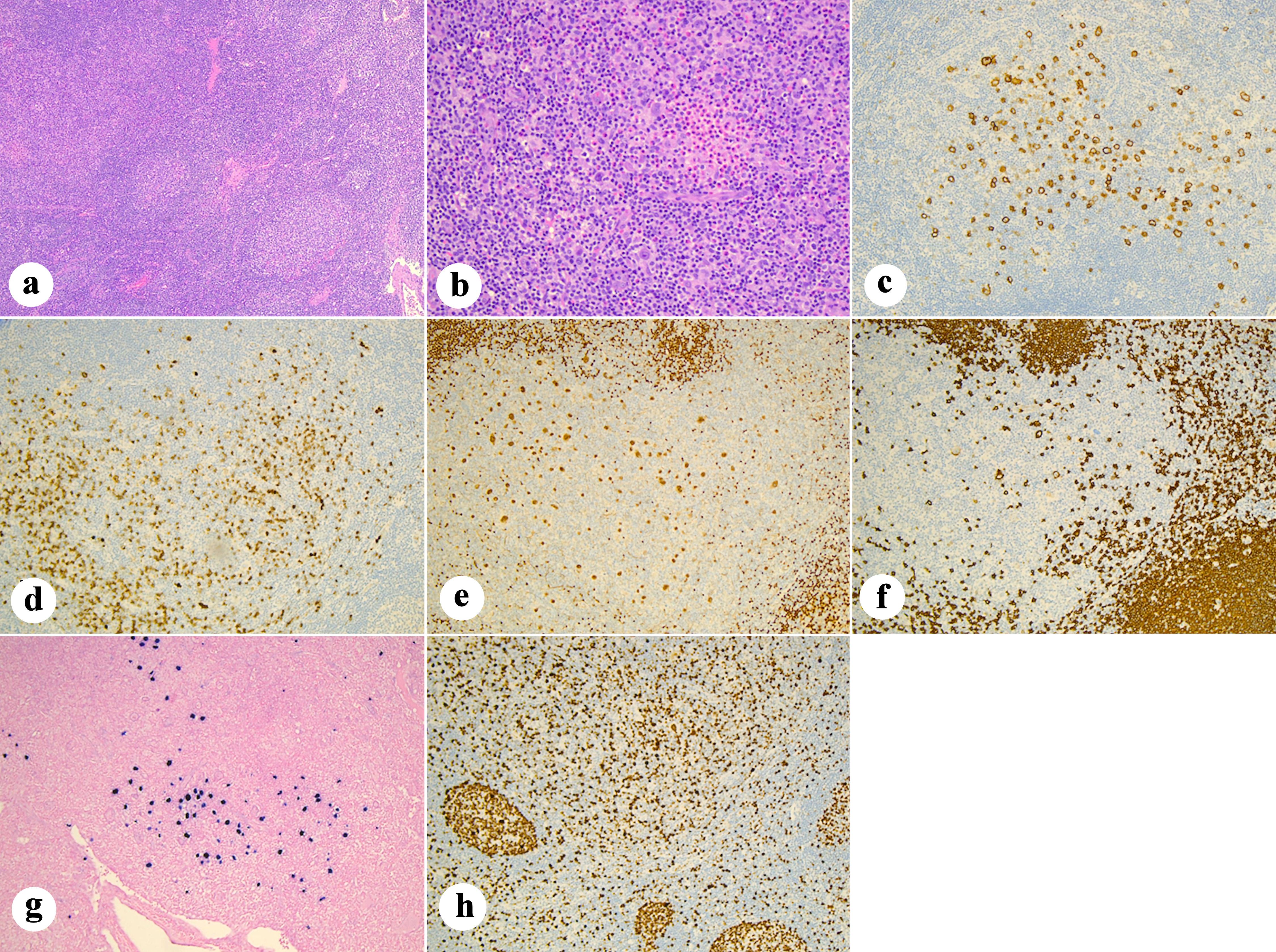
Dyskeratosis congenita (DC) is a rare inherited telomeropathy characterized by defective telomere maintenance and an elevated risk of hematologic malignancies. Classical Hodgkin lymphoma (cHL) is a rare malignancy described in patients with DC, and optimal treatment remains undefined due to overlapping toxicities of standard ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) and radiation therapy in this high-risk population. We present a 48-year-old man with longstanding thrombocytopenia who was diagnosed with DC based on clinical features and genetic testing. Two years post diagnosis, he developed stage IIA bulky cHL (nodular sclerosing, CD30+, Epstein-Barr virus (EBV)+). To mitigate pulmonary and myelotoxicity risks, he received a modified regimen of brentuximab vedotin (BV) combined with adriamycin, vinblastine, and dacarbazine (BV-AVD), with full omission of bleomycin. Treatment complications included peripheral neuropathy resulting in BV dose reduction and vinblastine discontinuation. Worsening thrombocytopenia led to discontinuation of dacarbazine. Interim imaging showed tumor regression, with post-treatment positron emission tomography with computed tomography (PET-CT) confirming complete metabolic response. Involved-site radiotherapy was omitted to minimize long-term risks of skin malignancy, local skin reactions and poor skin healing, in the context of DC. Post-treatment bone marrow evaluation showed no evidence of myeloid malignancy or lymphoma. This case demonstrates that modified BV-AVD can achieve complete metabolic remission in DC patients with cHL, while managing significant treatment-related toxicities. It underscores the critical need for individualized therapy in patients with DC and supports careful consideration of radiation omission to reduce secondary malignancy risk. These findings provide a potential therapeutic framework for managing Hodgkin lymphoma in patients with DC.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








