Immune Response Associated Hepatotoxicity in Hemophilia Gene Therapy: Mechanisms, Management, and Challenges
DOI:
https://doi.org/10.14740/jh2161Keywords:
AAV gene therapy, Hemophilia, Hepatotoxicity, Immune response activation, CorticosteroidsAbstract
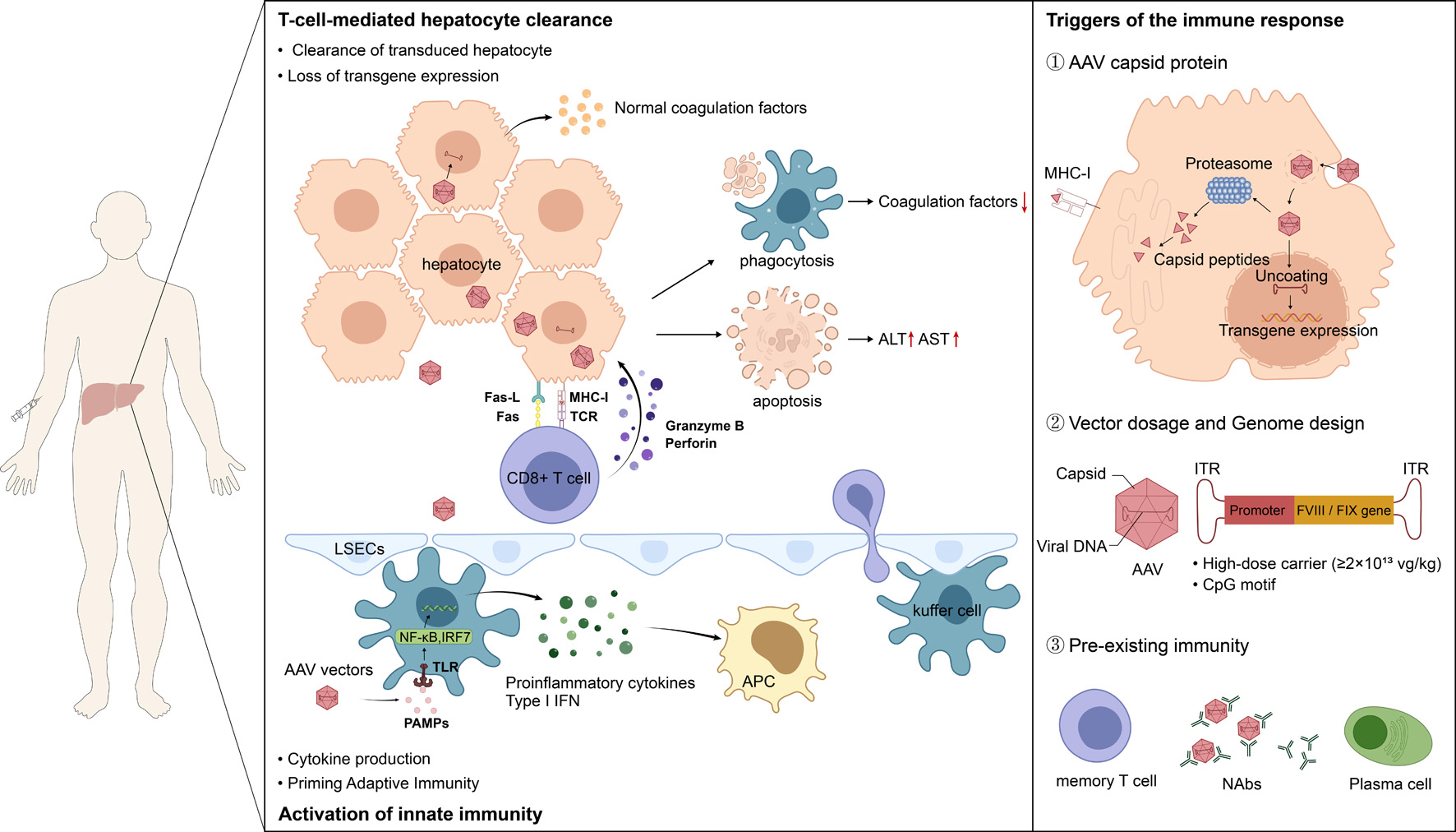
Adeno-associated virus (AAV)-based gene therapy offers the potential for long-term functional cure in patients with hemophilia A and B. However, immune responses triggered by the vector capsid or transgene product, leading to hepatotoxicity, represent a major challenge to the long-term stability of transgene expression and treatment safety. Based on reported clinical trials of hemophilia gene therapy, this review delves into the mechanisms of immune response activation following AAV gene therapy and summarizes the clinical features and monitoring strategies for hepatotoxicity, which primarily manifests as asymptomatic transaminase elevation. It highlights the roles of patient selection, vector optimization, and current clinical management strategies centered on corticosteroids in preventing and managing immune responses to stabilize transgene expression and prevent the decline of clotting factor levels. Furthermore, the review discusses potential differences between hemophilia A and B gene therapy, challenges such as long-term safety concerns (including tumorigenicity risk) and pre-existing immunity, and provides an outlook on future directions including vector engineering, immune modulation, and personalized treatment approaches. The aim is to offer practical insights for clinicians and promote the safer application of hemophilia gene therapy.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.








