| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Original Article
Volume 000, Number 000, March 2025, pages 000-000
Post-Induction Minimal Residual Disease in Pediatric Pre-B-Cell Acute Lymphoblastic Leukemia: A Step Towards Precision Medicine?
Ibrahim Ghemlasa, b, Ibrahim Al-Ebaida, Khawar Siddiquia, Sarah Ramiza, Saadiya Khana, Hawazen AlSaedia, Ali Al-Ahmaria, Abdullah Al-Jefria, Syed Jafria, Mouhab Ayasa
aDepartment of Pediatric Hematology/Oncology, King Faisal Specialist Hospital and Research Center, Riyadh, Saudi Arabia
bCorresponding Author: Ibrahim Ghemlas, Department of Pediatric Hematology/Oncology, King Faisal Specialist Hospital and Research Center, Riyadh 11211, Saudi Arabia
Manuscript submitted January 2, 2025, accepted February 10, 2025, published online March 3, 2025
Short title: Post-Induction MRD in Pediatric ALL
doi: https://doi.org/10.14740/jh1375
| Abstract | ▴Top |
Background: Acute lymphoblastic leukemia (ALL) is the most common pediatric malignancy with an incidence of 30% of pediatric cancers across the world and 34% amongst Saudi children with cancers. Minimal residual disease (MRD) is considered the most important independent predictor in determining the risk of relapse and long-term outcomes in ALL patients and plays a pivotal role in guiding risk-adapted therapies. The aim of this research was to study the role of MRD on survival benefits in our patient population.
Methods: We reviewed medical records of 108 pediatric (age ≤ 14 years) ALL patients treated between January 2016 and December 2018 at our center to assess if MRD and other associated risk factors affect the outcome of patients at post-induction and post-consolidation phases of the treatment protocols.
Results: The median follow-up time in our cohort of patients was 75.6 months (95% confidence interval: 71.3 - 79.8 months). With a mortality rate of 10.2% (11 deaths out of 108 cases), overall survival (OS) of the whole cohort was 89.2±3.1%. OS was significantly lower in post-induction MRD-positive cases than in MRD-negative cases (74.2±8.6% vs. 94.7±2.6%, P = 0.006). It was worse among those patients who underwent consolidation therapy and had positive post-consolidation MRD. Event-free survival (EFS) was also significantly poor in post-induction MRD-positive cases (61.1±10.2% vs. 92.1±3.1%, P = 0.001). Twenty-seven patients who received consolidation therapy had the poorest EFS (P = 0.031). Amongst all the factors, including age at diagnosis, gender, white blood cell count, central nervous system status, risk group or cytogenetics, only post-induction MRD positivity was found to be significantly associated with OS.
Conclusion: Post-induction MRD is one of the most important factors affecting the patient’s outcome. Post-induction MRD-positive patients fared better after receiving consolidation therapy. No significant association was found between post-induction MRD and other risk factors.
Keywords: Minimum residual disease; Acute lymphoblastic leukemia; Stem cell transplantation; Pediatric; Bone marrow; Risk factors; Survival
| Introduction | ▴Top |
Acute lymphoblastic leukemia (ALL) is the most common pediatric malignancy with an incidence of 30% of pediatric cancers across the world and 34% amongst Saudi children with cancers [1, 2]. Pediatric ALL has an overall favorable prognosis, which is mainly attributed to the recent advancement in diagnosis and treatment. Over the last few decades, the incorporation of minimal residual disease (MRD) in the ALL treatment has played a significant role in predicting the patient’s response, risk stratification and tailoring the treatment [3, 4]. An essential part of ALL therapy is to monitor the response to treatment by periodic examination of bone marrow aspirates. Due to the similarities in morphology of leukemic cells and the normal lymphohematopoietic progenitors, it becomes difficult to ascertain the degree of leukemia in the bone marrow aspirates, leading some patients to still harbor significant leukemia and yet receive a less intensive post-remission therapy, or to treatment intensifications and toxicities for patients due to an erroneous overestimation of the disease [5]. Accordingly, there has been a considerable effort to develop methods, which could identify the degree of residual leukemia in patients considered to be in morphologic remission, by measuring MRD.
MRD detection in ALL was first used in 1980s by using immunofluorescence microscope [6]. In 1990s, the prognostic value and clinical significance of MRD in ALL was investigated in various centers across Europe and the United States [7]. Since MRD assessments are time-point specific, their role in the prognosis of very high-risk B-cell ALL patients is extremely critical. Patients who fail to achieve end of induction MRD clearance tend to have poor outcomes [3, 8]. Conversely, patients who achieve very early MRD clearance fare well [9]. Various studies have established a causal relationship between MRD and risk of relapse, signifying its prognostic value [10, 11]. The use of MRD assessments in pediatric ALL is relatively new in Saudi Arabia and hence there is a dearth of data. The primary objective of this review was to study the impact of MRD assessments on our patients following induction and consolidation and the association of other factors, which could play a role in determining their outcome.
| Materials and Methods | ▴Top |
Study design
This retrospective analysis was carried out on 108 pediatric non-infantile patients (age at diagnosis < 14 years) who were diagnosed with and treated for pre-B ALL at our institution. The time fame for the patients’ enrollment into the study was from January 2016 to December 2018. Data on clinical characteristics, hematopoietic profile, risk stratification, treatment and outcome were collected from the patients’ electronic medical records and prospectively maintained institutional research databases. Primary measure was to review the outcome of all patients in the cohort in terms of disease status with reference to MRD (> 0.01%) after completing the induction chemotherapy. Secondary measure was to review the outcome of post-induction MRD-positive patients after completing the consolidation treatment as well as the role of any associated risk factors in determining the patient’s outcome. Being a retrospective research study, a waiver of parental informed consent was obtained from the Institutional Review Board (IRB) of the hospital.
End point definitions
MRD positivity was taken as more than 0.01% using eight-color flow cytometry (FCM). Relapse of primary disease and death from any cause was taken as an event.
Treatment protocols
Based on risk stratification, two Children’s Oncology Group (COG) protocols were used in treating our patients. The standard risk protocol (AALL0331) is a three-drug protocol consisting of vincristine and asparaginase with steroids. The high risk is a four-drug protocol (AALL0232), comprising vincristine, asparaginase and daunorubicin with steroids. For refractory and relapsed group of patients, salvage therapy in the form of clofarabine, etoposide, cyclophosphamide and intrathecal chemotherapy was used.
Cytogenetics
Cytogenetics played a pivotal role in assigning the risk groups of our cohort. For the favorable group, patients who had either/or hyperdiploidy, ETV6-RUNXI, NUMT1 rearrangement, translocation (1;19); TCF3-PBX1 and translocation (5;14) IGH/IL3 were considered. Unfavorable cytogenetics were represented by iAMP21, KMT2A, MLL gene rearrangement, hypodiploidy (n < 44 chromosomes and/or a DNA index < 0.81) and translocation BCR-ABL.
Statistical analysis
All continuous data are presented as median with minimum and maximum points. Independent-samples Mann-Whitney U test was used to test the significance of difference among the continuous variables. Chi-square test or Fisher’s exact test was employed to test the significance of association between categorical variables. Kaplan-Meier curves were drawn for survival analyses. IBM-SPSS for Windows (version 20.0) was used for statistical analysis of the data. A P-value less than 0.05 was considered as significant.
Ethical considerations
The proposal of this research study was submitted to the IRB before initiation and was approved by the Research Advisory Committee through established procedures via approval number 2221184.
Ethical compliance with human/animal study
This study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
| Results | ▴Top |
Almost half of the patients were male (n = 52, 48.1%) and median age of the group at diagnosis was 4.3 years (range, 1.1 - 13.9 years). Details on characteristics are provided in Table 1. Majority of the patients were under 10 years of age at diagnosis (n = 91, 84.3%), with white blood cell (WBC) at presentation less than 50 × 109/L (n = 91, 84.3%) and had central nervous system (CNS)-1 status (n = 91, 84.3%). Disease was of high risk as per National Cancer Institute (NCI) risk stratification in 42 (38.9%). A variety of treatment protocols were employed for the first-line treatment with AALL0331 being the most commonly used (n = 64, 9.3%). Family history of cancer was positive in eight (7.4%) cases, three (37.5%) had documented leukemia and the remaining five (62.5%) were listed as non-hematolymphoid malignancy; one family had a history of colon cancer and for the remaining exact diagnosis was not available. Whereas one (0.9%) patient had global developmental delay. Three (2.8%) patients had Down’s syndrome and sickle cell anemia and mitochondrial disease were recorded in one (0.9%) each. CNS symptoms were positive in six patients (5.6%). BCR/ABL was positive in eight cases (8%, out of 100 done), TEL/AML fusion gene in 60 (60%, out of 100 done) and MLL gene rearrangement in three (3%, out of 100 done). Cytogenetic studies were unfavorable in eight cases (8%, out of 100 done).
 Click to view | Table 1. Clinical Characteristics of the Patients |
Post-induction MRD was found to be positive in 30 cases (27.8%). It was not significantly associated with any of the age at diagnosis, gender, WBC, CNS status, risk group or cytogenetics (Table 2). Twenty-seven (90.0%) of these 30 cases with positive MRD completed consolidation phase with 23 (85.2%) receiving high-risk protocol, two (7.4%) very high-risk protocol and the remaining two (7.4%) got salvage chemotherapy consisting of clofarabine, etoposide, cyclophosphamide and intrathecal chemotherapy. Post-consolidation MRD was positive in seven (25.9%) of these 27 cases. It was also not found to be significantly associated with age at diagnosis, gender, WBC, CNS status, risk group or cytogenetics (Table 3). Relapse rate in the sub-group of 27 cases was 22.2% (n = 6); two (33.3%) were from MRD-positive and remaining four (66.7%) from MRD-negative group of cases (Fig. 1). In four post-induction MRD-negative cases, median time to relapse from diagnosis was 16.1 months. Median time to relapse from diagnosis in post-consolidation MRD-positive cases was 9.8 months compared to 51.4 months in post-consolidation MRD-negative cases (P = 0.133, Table 4). Cumulative incidence of relapse (CIR) at 5 years from diagnosis was 7.9% for the whole cohort. In post-induction MRD-positive cases, it was 14.9% compared to 5.4% in their counterparts. Testicular disease was found in only one patient, who was diagnosed at the age of 8.4 years, CNS-1 and high risk. He had negative post-induction MRD and was alive in continued remission without any relapse of primary disease with a follow-up of 6.4 years since diagnosis.
 Click to view | Table 2. Post-Induction MRD With Clinical and Biological Risk Factors (n = 108) |
 Click to view | Table 3. Post-Consolidation MRD With Clinical and Biological Risk Factors (n = 27) |
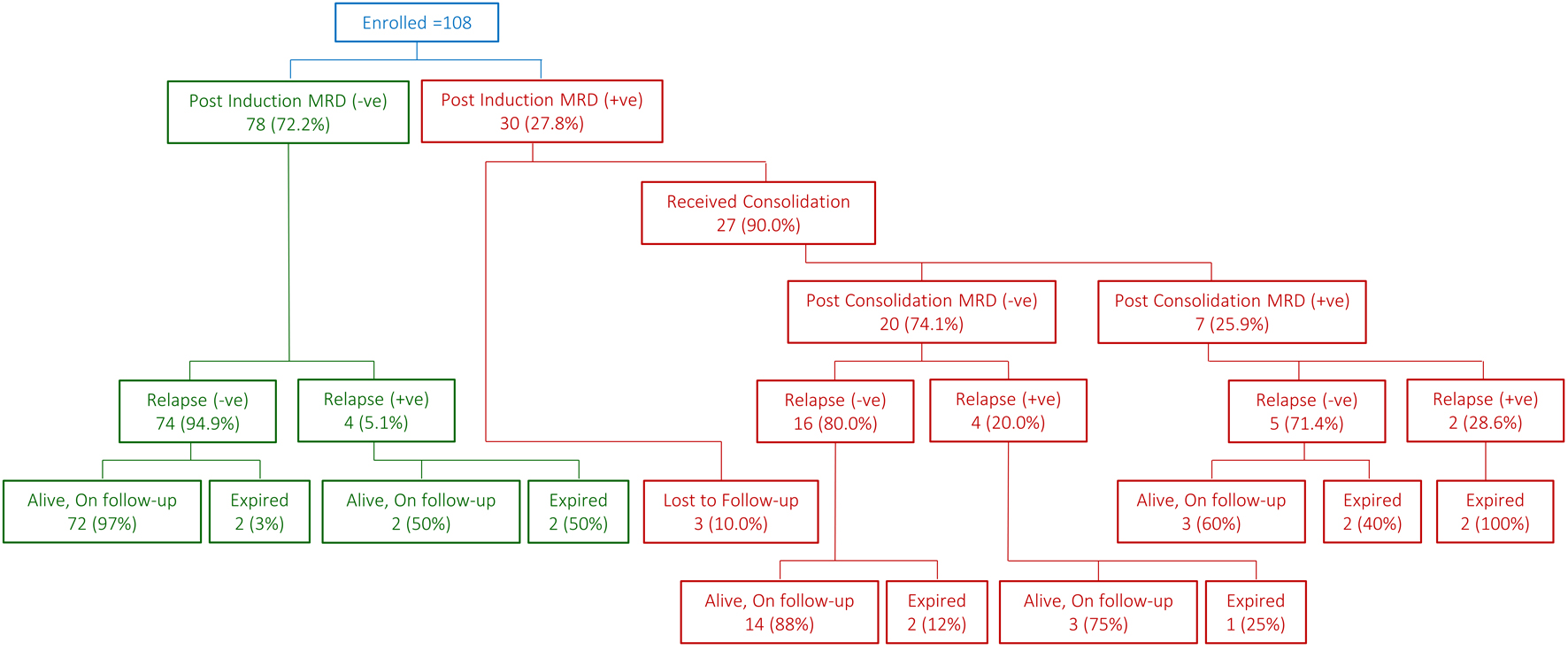 Click for large image | Figure 1. Snapshot of patient flow. |
 Click to view | Table 4. Time and Site of Relapse |
In univariate analysis, post-induction MRD positivity was found to be significantly associated with relapse of the disease (odds ratio: 4.6, 95% confidence interval (CI): 1.2 - 17.8, P = 0.026) and mortality (odds ratio: 5.6, 95% CI: 1.5 - 21.0, P = 0.010).
Median follow-up time in our cohort of patients was 75.6 months (95% CI: 71.3 - 79.8 months). With a mortality rate of 10.2% (11 deaths out of 108 cases), overall survival (OS) of the whole cohort was 89.2±3.1%. OS was significantly poor in post-induction MRD-positive cases than in MRD-negative cases (74.2±8.6% vs. 94.7±2.6%, P = 0.006, Table 5, Fig. 2). It was inferior among those patients who underwent consolidation therapy and had positive post-consolidation MRD (42.9±18.7% vs. 82.2±9.4%, P = 0.014, Fig. 3). Event-free survival (EFS) was also significantly poor in post-induction MRD-positive cases (61.1±10.2% vs. 92.1±3.1%, P = 0.001, Table 5). The same was also true for those 27 cases who received consolidation therapy (P = 0.031, Table 5, Figs. 4 and 5). In a multivariable setting while controlling for gender and risk group, post-induction MRD positivity only was found to be significantly associated with OS (hazard ratio: 5.2, 95% CI: 1.5 - 18.0, P = 0.009). Small sample size for the sub-group of 27 patients who received post-consolidation therapy precluded the multivariable analysis.
 Click to view | Table 5. Overall and Event-Free Survival |
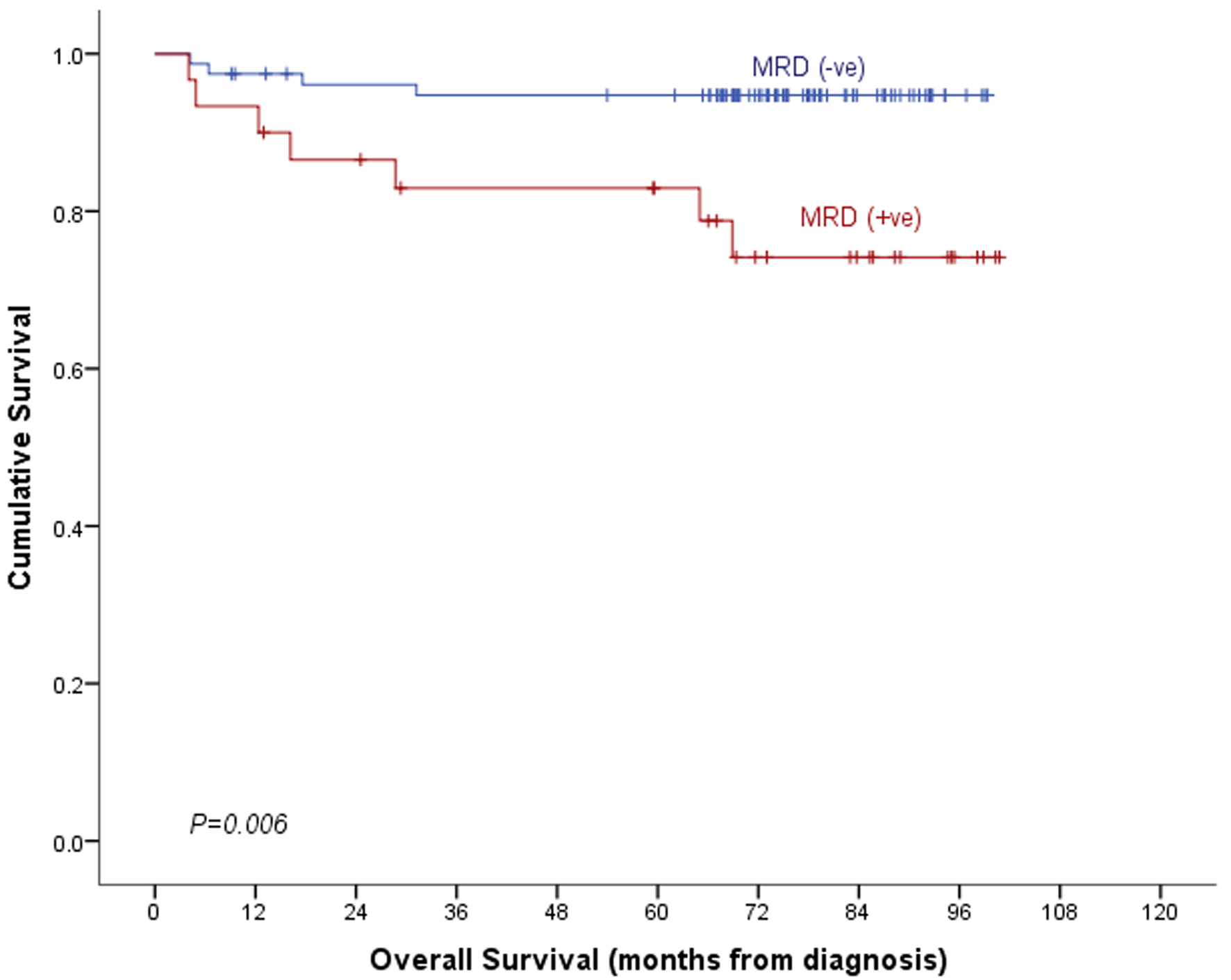 Click for large image | Figure 2. Overall survival by post-induction MRD. MRD: minimal residual disease. |
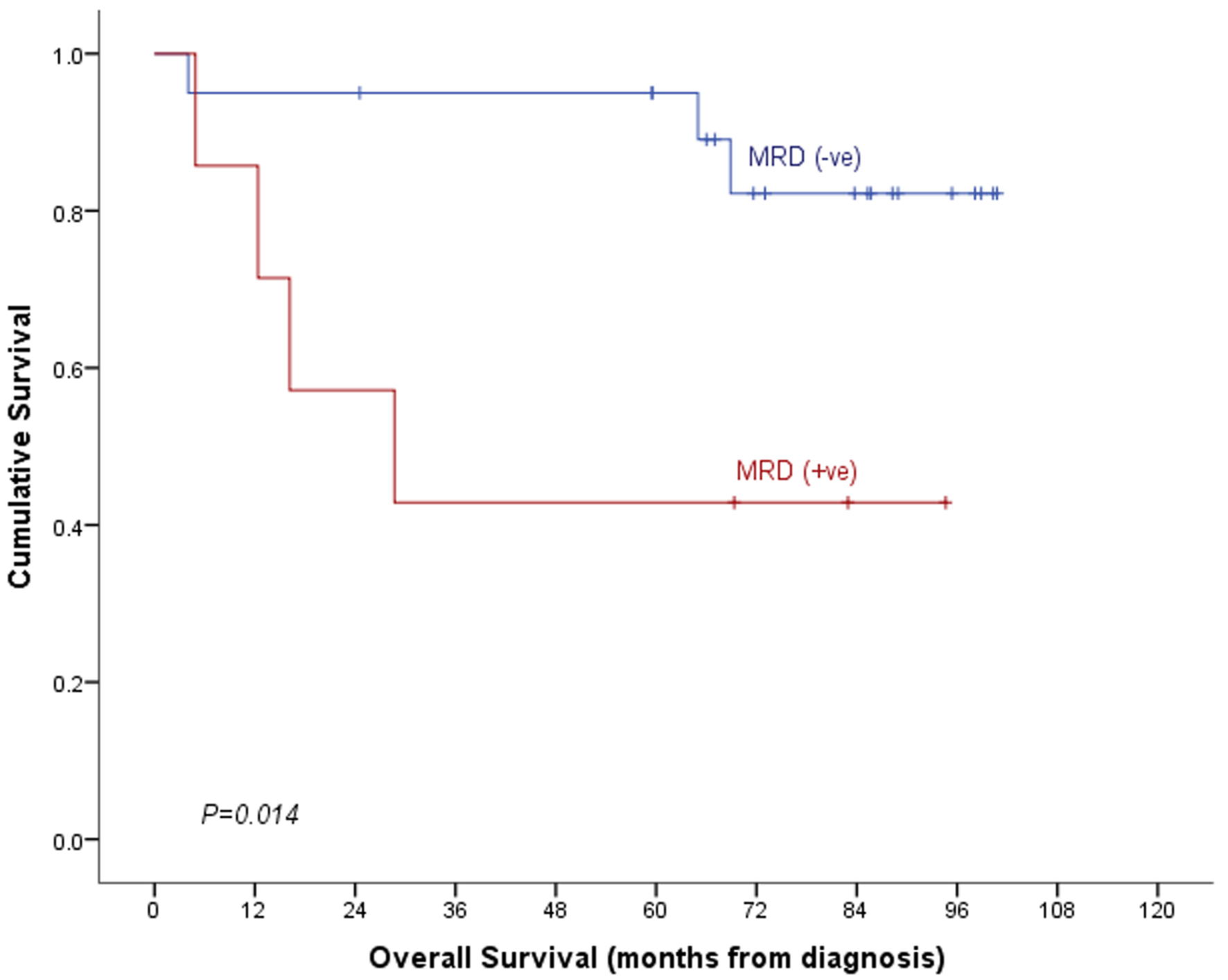 Click for large image | Figure 3. Overall survival by post-consolidation MRD. MRD: minimal residual disease. |
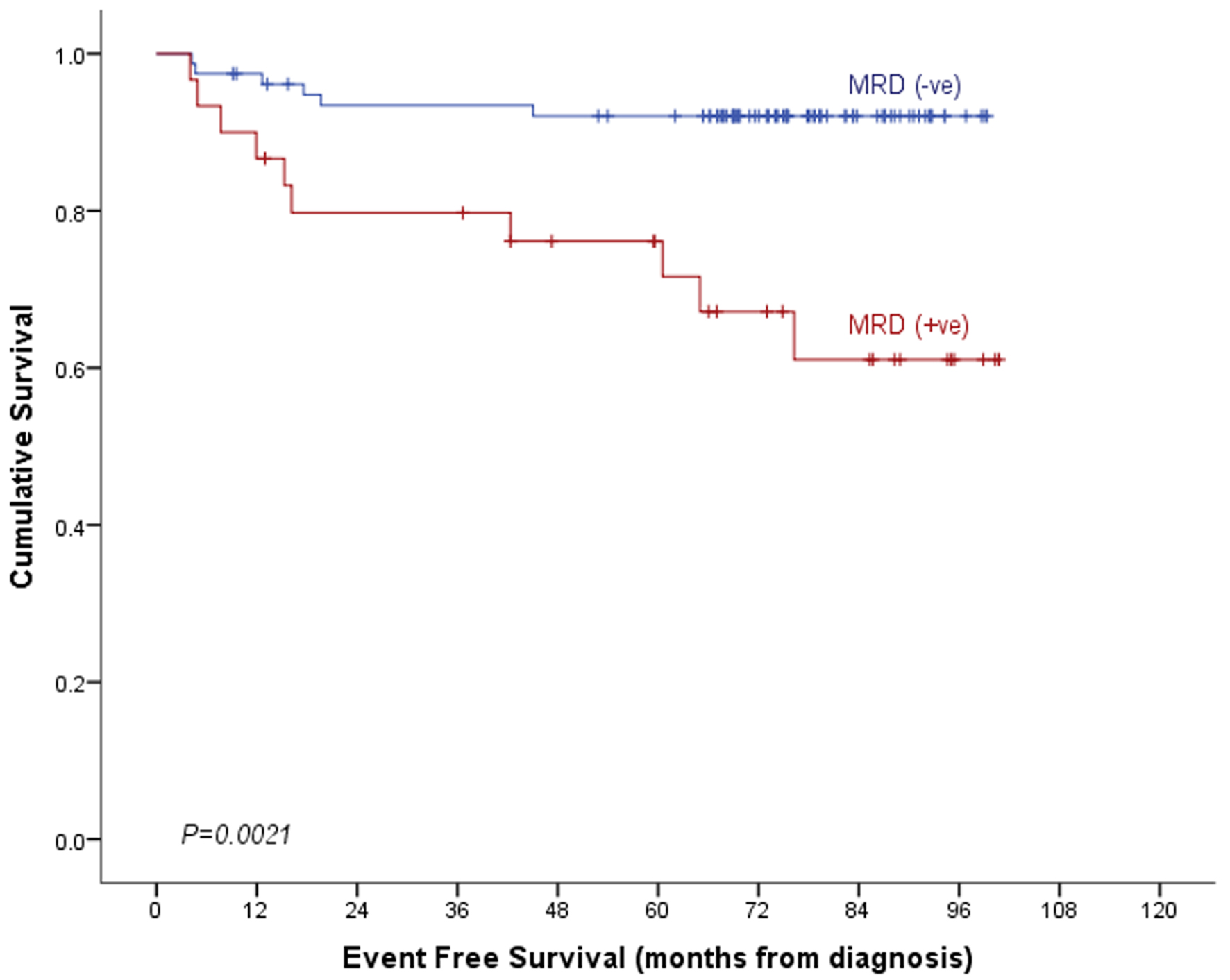 Click for large image | Figure 4. Event-free survival by post-induction MRD. MRD: minimal residual disease. |
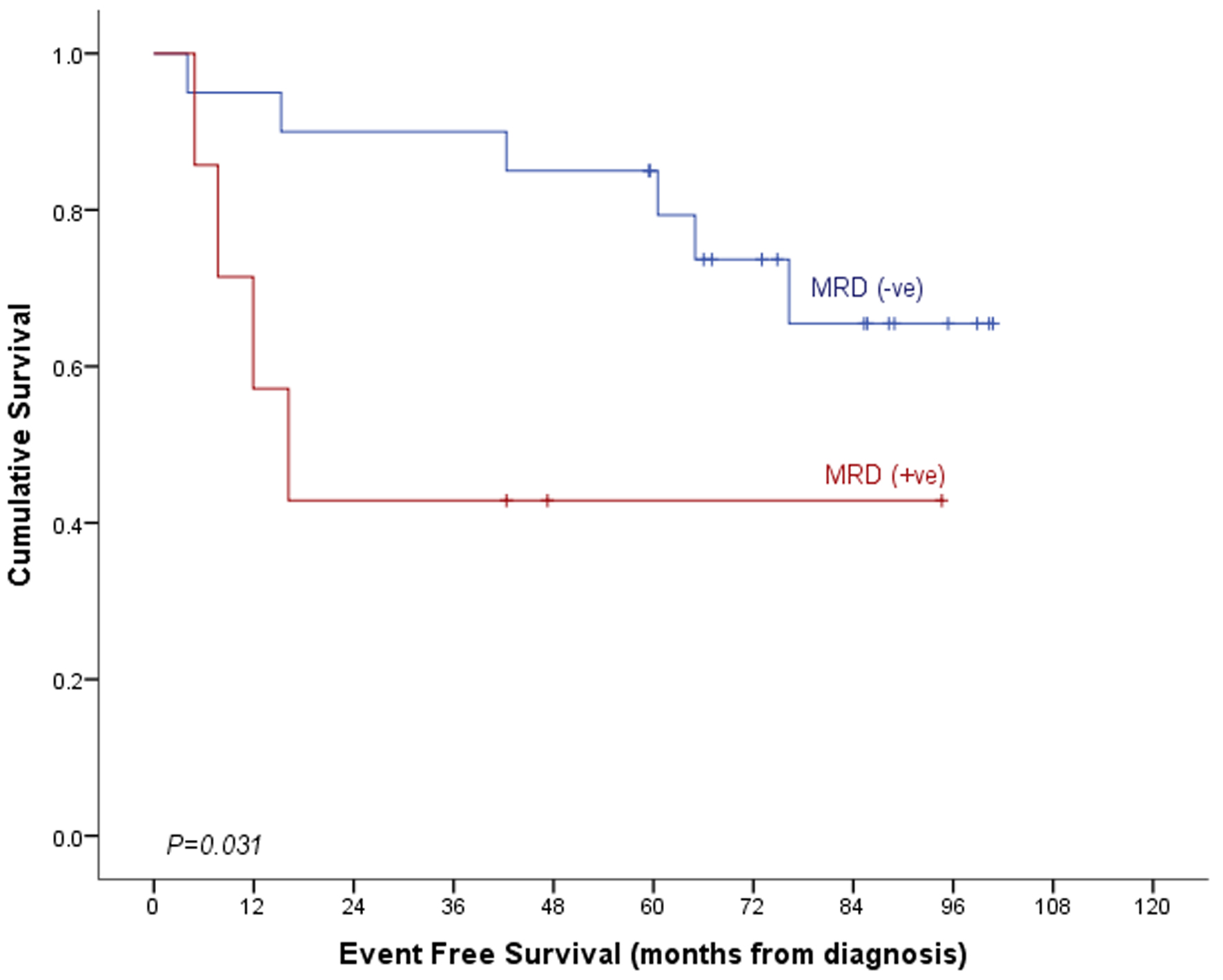 Click for large image | Figure 5. Event-free survival by post-consolidation MRD. MRD: minimal residual disease. |
| Discussion | ▴Top |
The treatment of ALL in children has made huge strides during the last few decades, inducing a complete remission, based on cytomorphological criteria, in almost 95-98% patients. However, about 25-30% of these patients do relapse, suggesting that leukemia was not completely eradicated and hence questioning the reliability of cytomorphological criteria for adequate assessment of the remission [12]. Therefore, more sensitive techniques are needed to confirm the treatment efficacy and to enhance risk stratification of the treatment protocols. Identification of MRD in ALL patients, following chemotherapy, has emerged as one of the most important tools for guiding the prognosis and treatment choices [13]. The two most significant and reliable methods used in clinical practice to assess MRD in ALL are FCM analysis of leukemia-associated immunophenotypes and polymerase chain reaction (PCR) amplification of antigen-receptor gene rearrangements [14]. MRD has been shown to be particularly prognostic during the early phases of treatment, such as during or after induction and early in consolidation, which helps in risk stratification and shaping the appropriate treatment [15]. In addition to MRD, there are additional factors which have been known to influence the outcome of ALL pediatric patients, such as age and leukocyte count at diagnosis [16]. There have been a few studies claiming that the MRD is prognostically superior even after adjusting for those risk factors [17], although the data are too scarce to fully establish a relationship between MRD and other risk factors in predicting the outcome.
The use of MRD in predicting the outcome of pediatric ALL in Saudi Arabia is relatively new and hence there is a dearth of data on it. We did not observe any significant difference in the MRD between patients belonging to standard or high-risk groups for our patient population. Our post-induction MRD-negative patients fared well with an excellent OS and EFS rate compared to the MRD-positive patients (Figs. 1, 2 and 4). A Nordic study by Nyvold et al on 104 patients produced the similar results [18].
Patients who were MRD-positive at the end of consolidation therapy had the worse prognosis, with a poor OS and EFS rate (Figs. 1, 3 and 5) which is in concordance with a COG study where they found that a post-consolidation MRD-positive disease was associated with a 5-year EFS of 43% [19]. MRD following induction and consolidation has a significant impact on shaping the treatment plan and on the subsequent outcome. A Dutch study by van Binsbergen et al, on high-risk ALL patients (no complete remission, MRD > 10-3 after consolidation) who received one induction and consolidation followed by three high-risk chemotherapy blocks, followed by either stem cell transplantation or further chemotherapy, found that patients with either negative MRD levels or low-positive MRD during high-risk chemotherapy had a significantly lower 5-year CIR of 2.2% compared with the one with high-positive MRD levels (CIR of 15.4%) [20]. An Italian study observed that patients who tested high-positive, low-positive, or negative, had CIR of 83.3%, 34.8%, and 8.6% respectively. About two-thirds of these positive cases were identified within 4 months after induction-consolidation phases, endorsing the significance of MRD following induction and consolidation [21].
Another important feature of post-induction and consolidation MRD is its impact on the cost effectiveness of the treatment in ALL patients. A Canadian study concluded that MRD testing in newly diagnosed ALL patients decreased the burden on the health care system, by guiding towards risk-directed therapies [22].
Conclusion
The role of MRD diagnostics and its applications are yet to be established in Saudi Arabia. In our cohort of patients, post-induction MRD-positive patients fared better after receiving consolidation therapy. There were no significant associations found between post-induction MRD and other risk factors.
Acknowledgments
None to declare.
Financial Disclosure
This work did not receive any financial support in any form from any funding agency.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
Data of interest collected from the patients’ medical records were secured as governed by the institutional policies on patient confidentiality and privacy. No informed consents were obtained since this was a retrospective study and all data items collected were already documented in medical charts as part of the patient care and disease management documentation.
Author Contributions
All authors certify that they have participated sufficiently in the intellectual content and the analysis of data. Each author has reviewed the final version of the manuscript and approves it for publication. Should the editors request the data upon which the work is based, the authors shall produce it. IG and IAE conceived the idea, KS designed the study, prepared the database, analyzed the data and contributed towards results reporting and manuscript preparation and review. IAE collected the data and prepared the manuscript. IG supervised the study progression. SR, SK, HAS, AAA, AAJ, MA and IG reviewed the results and critically inspected the manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
ALL: acute lymphoblastic leukemia; ANC: absolute neutrophil count; CIR: cumulative incidence of relapse; CNS: central nervous system; COG: Children’s Oncology Group; EFS: event-free survival; FCM: flow cytometry; HR: high risk; IRB: Institutional Review Board; MRD: minimal residual disease; NCI: National Cancer Institute; OS: overall survival; PCR: polymerase chain reaction; SD: standard deviation; WBC: white blood cell
| References | ▴Top |
- Bartram CR, Schrauder A, Kohler R, Schrappe M. Acute lymphoblastic leukemia in children: treatment planning via minimal residual disease assessment. Dtsch Arztebl Int. 2012;109(40):652-658.
doi pubmed - Al-Mutlaq HM, Bawazir AA, Jradi H, Al-Dhalaan ZA, Al-Shehri A. Patterns of childhood cancer incidence in Saudi Arabia (1999- 2008). Asian Pac J Cancer Prev. 2015;16(2):431-435.
doi pubmed - Mortuza FY, Papaioannou M, Moreira IM, Coyle LA, Gameiro P, Gandini D, Prentice HG, et al. Minimal residual disease tests provide an independent predictor of clinical outcome in adult acute lymphoblastic leukemia. J Clin Oncol. 2002;20(4):1094-1104.
doi pubmed - Menon H, Singh PK, Bagal B, Dolai T, Jain A, Chaudhri A. Minimal residual disease in the management of B-cell acute lymphoblastic leukemia: a systematic review of studies from Indian settings. Indian J Hematol Blood Transfus. 2024;40(1):1-11.
doi pubmed - Campana D. Status of minimal residual disease testing in childhood haematological malignancies. Br J Haematol. 2008;143(4):481-489.
doi pubmed - Bradstock KF, Janossy G, Tidman N, Papageorgiou ES, Prentice HG, Willoughby M, Hoffbrand AV. Immunological monitoring of residual disease in treated thymic acute lymphoblastic leukaemia. Leuk Res. 1981;5(4-5):301-309.
doi pubmed - Kruse A, Abdel-Azim N, Kim HN, Ruan Y, Phan V, Ogana H, Wang W, et al. Minimal residual disease detection in acute lymphoblastic leukemia. Int J Mol Sci. 2020;21(3):1054.
doi pubmed - Salzer WL, Burke MJ, Devidas M, Dai Y, Heerema NA, Carroll AJ, Gore L, et al. Minimal residual disease at end of induction and consolidation remain important prognostic indicators for newly diagnosed children and young adults with very high-risk (VHR) B-lymphoblastic leukemia (B-ALL): Children’s Oncology Group AALL1131. JCO. 2021;39:10004-10104.
doi - Lee S, Kim DW, Cho BS, Yoon JH, Shin SH, Yahng SA, Lee SE, et al. Impact of minimal residual disease kinetics during imatinib-based treatment on transplantation outcome in Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia. 2012;26(11):2367-2374.
doi pubmed - Patil PP, Jafa E, Aggarwal M. Minimal residual disease in acute lymphoblastic leukemia. Indian J Med Paediatr Oncol. 2021;42:71-76.
doi - Berry DA, Zhou S, Higley H, Mukundan L, Fu S, Reaman GH, Wood BL, et al. Association of minimal residual disease with clinical outcome in pediatric and adult acute lymphoblastic leukemia: a meta-analysis. JAMA Oncol. 2017;3(7):e170580.
doi pubmed - van Dongen JJ, Seriu T, Panzer-Grumayer ER, Biondi A, Pongers-Willemse MJ, Corral L, Stolz F, et al. Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood. Lancet. 1998;352(9142):1731-1738.
doi pubmed - Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J, Bakkus M, et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia. European Organization for Research and Treatment of Cancer—Childhood Leukemia Cooperative Group. N Engl J Med. 1998;339(9):591-598.
doi pubmed - Gaipa G, Basso G, Biondi A, Campana D. Detection of minimal residual disease in pediatric acute lymphoblastic leukemia. Cytometry B Clin Cytom. 2013;84(6):359-369.
doi pubmed - Coustan-Smith E, Sancho J, Behm FG, Hancock ML, Razzouk BI, Ribeiro RC, Rivera GK, et al. Prognostic importance of measuring early clearance of leukemic cells by flow cytometry in childhood acute lymphoblastic leukemia. Blood. 2002;100(1):52-58.
doi pubmed - Silverman LB, Sallan SE. Newly diagnosed childhood acute lymphoblastic leukemia: update on prognostic factors and treatment. Curr Opin Hematol. 2003;10(4):290-296.
doi pubmed - Coustan-Smith E, Sancho J, Hancock ML, Boyett JM, Behm FG, Raimondi SC, Sandlund JT, et al. Clinical importance of minimal residual disease in childhood acute lymphoblastic leukemia. Blood. 2000;96(8):2691-2696.
pubmed - Nyvold C, Madsen HO, Ryder LP, Seyfarth J, Svejgaard A, Clausen N, Wesenberg F, et al. Precise quantification of minimal residual disease at day 29 allows identification of children with acute lymphoblastic leukemia and an excellent outcome. Blood. 2002;99(4):1253-1258.
doi pubmed - Borowitz MJ, Devidas M, Hunger SP, Bowman WP, Carroll AJ, Carroll WL, Linda S, et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia and its relationship to other prognostic factors: a Children's Oncology Group study. Blood. 2008;111(12):5477-5485.
doi pubmed - van Binsbergen AL, de Haas V, van der Velden VHJ, de Groot-Kruseman HA, Fiocco MF, Pieters R. Efficacy and toxicity of high-risk therapy of the Dutch Childhood Oncology Group in childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. 2022;69(2):e29387.
doi pubmed - Paganin M, Fabbri G, Conter V, Barisone E, Polato K, Cazzaniga G, Giraldi E, et al. Postinduction minimal residual disease monitoring by polymerase chain reaction in children with acute lymphoblastic leukemia. J Clin Oncol. 2014;32(31):3553-3558.
doi pubmed - Health Quality Ontario. Minimal residual disease evaluation in childhood acute lymphoblastic leukemia: an economic analysis. Ont Health Technol Assess Ser. 2016;16(8):1-83.
pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.