| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Case Report
Volume 000, Number 000, February 2025, pages 000-000
Aggressive Extramedullary Multiple Myeloma Presenting as Small Bowel Obstruction
Paul J. Wurtza, e, Kevin McGovernb, Jamie Shahc, Kristin E. Stolld, Devin Moorea
aDepartment of Internal Medicine, Brooke Army Medical Center, Fort Sam Houston, TX 78234, USA
bDepartment of General Surgery, Brooke Army Medical Center, Fort Sam Houston, TX 78234, USA
cDepartment of Pathology, Brooke Army Medical Center, Fort Sam Houston, TX 78234, USA
dDepartment of Hematology and Oncology, Brooke Army Medical Center, Fort Sam Houston, TX 78234, USA
eCorresponding Author: Paul J. Wurtz, Department of Internal Medicine, Brooke Army Medical Center, Fort Sam Houston, TX 78234, USA
Manuscript submitted January 16, 2025, accepted February 12, 2025, published online February 25, 2025
Short title: Aggressive Extramedullary Multiple Myeloma
doi: https://doi.org/10.14740/jh2031
| Abstract | ▴Top |
De novo extramedullary multiple myeloma (EMM) is a rare subset of multiple myeloma (MM) defined by the presence of clonal plasma cells (PC) outside of the bone marrow. It is associated with refractory disease and adverse outcomes. Even in EMM, plasmacytomas within the duodenum, jejunum, and ileum are uncommon, with fewer than 70 cases reported in the literature. Here, we present a particularly aggressive case of EMM resulting in a small bowel obstruction secondary to an intraluminal plasmacytoma while on myeloma-directed therapy. The patient underwent surgical resection with anastomosis and was transitioned to more definitive cytotoxic chemotherapy followed by autologous stem cell rescue. This case highlights challenges in the management of EMM over standard MM and argues that dedicated clinical trials for patients with aggressive EMM are warranted to further understand the unique pathophysiology and improve overall survival.
Keywords: Extramedullary multiple myeloma; Plasmacytoma; Small bowel obstruction; Hepatic plasmacytoma; Iron deficiency anemia
| Introduction | ▴Top |
Multiple myeloma (MM) is a common hematologic malignancy characterized by a monoclonal proliferation of plasma cells within the bone marrow [1]. A subset of MM, known as extramedullary multiple myeloma (EMM), is defined by the presence of plasma cells outside the bone marrow due to plasma cells losing their bone marrow homing machinery [2]. De novo EMM is diagnosed when extramedullary disease is present at initial diagnosis, and it is appreciated in 6-8% of new MM diagnoses [3]. De novo EMM is associated with refractory disease and adverse clinical outcomes [4].
When clonal plasma cells spread outside the protective environment of the bone marrow, they gain the ability to proliferate more freely, contributing to the aggressive nature of the disease. These plasma cells can spread to extramedullary sites, such as the liver, skin, kidneys, and pancreas, which indicates a shift from typical MM behavior and a higher likelihood of resistance to therapy [5, 6]. The resulting soft tissue plasmacytomas can grow quickly in their nonnative tissues, resulting in various complications depending on their location.
Extramedullary disease is often identified through specific genetic and molecular profiles, which are associated with the underlying pathophysiology of disease progression beyond the bone marrow. These include mutations in tumor suppressor genes such as p53 in addition to the activation of cell adhesion pathways like focal adhesion kinase (FAK). Fluorescent in situ hybridization (FISH) characteristics of an EMM population revealed a high prevalence of 17p deletions, 1q duplications, CCND1/IGH translocations, and complex karyotypes, which likely contribute to treatment challenges [2, 7-10].
Biologic characteristics of EMM include an immature or plasmablastic morphology, often resembling that of advanced disease in MM, including signs of bone marrow suppression such as anemia and thrombocytopenia. Other markers are similar to those of transformed malignant lymphoma and include a high lactate dehydrogenase (LDH) level and multi-organ involvement [11]. Even in EMM, plasmacytomas within the duodenum, jejunum, and ileum are uncommon, with fewer than 70 cases reported in the literature.
Many of the standard treatment options that are used to treat MM are also available to treat EMM, but they are without the robust literature support. The regimen consisting of daratumumab, bortezomib, cyclophosphamide, and dexamethasone (DaraVCD) is currently being studied as a potential option, with some success [4]. Many of the treatments that are used are similar to the medications used in traditional MM, such as proteasome inhibitors, immunomodulatory drugs (IMiDs), and autologous stem cell rescue (auto-SCR). Novel treatment options have been proposed for refractory disease, such as combinations of the above therapies in addition to tandem auto-SCR, bidirectional T-cell engager (BiTE) therapy, and chimeric antigen receptor T-cell (CAR-T) therapy [12]. Imaging strategies also differ in EMM, where positron emission tomography-computed tomography (PET-CT) has proven valuable for detecting extramedullary lesions and monitoring treatment response, even when serum markers fail to indicate disease progression. Yet, PET-CT is not part of the current standard response criteria for MM, highlighting an area where further guidelines and research are needed to ensure appropriate management of this unique disease entity [5]. We describe a case of a middle-aged woman presenting with extensive extramedullary disease, in which many of the complexities of EMM are highlighted, including the diagnostics, treatment, and monitoring options.
| Case Report | ▴Top |
A 58-year-old woman was diagnosed with R-ISS stage III MM with extensive extramedullary involvement after presenting with a thoracic plasmacytoma resulting in cord compression. Given the potential for rapid neurological deterioration, this plasmacytoma necessitated urgent intervention to prevent lasting functional deficits. The full extent of her disease became evident with further imaging, which revealed additional extramedullary plasmacytomas located in the left choroid, left cerebral hemisphere dura, and the soft tissues surrounding the right orbit.
Chromosome analysis (Fig. 1) [1, 7, 8] and FISH (Fig. 2) from her diagnostic bone marrow biopsy showed a complex karyotype, featuring a tetrasomy and eight trisomies (many of which are not typically associated with MM), along with a notably aggressive p53 mutation.
 Click for large image | Figure 1. Cytogenetics results. Findings with an asterisk (*) have an established association with multiple myeloma [1, 7, 8]. N/A: not applicable; Chr: chromosome. |
 Click for large image | Figure 2. Fluorescence in situ hybridization (FISH) results. |
She was started on first-line therapy with standard-dose bortezomib, lenalidomide, and dexamethasone (RVD). After 4 months of treatment, she achieved a very good partial response (VGPR), evidenced by complete serologic remission, the absence of an M-spike on immunofixation electrophoresis (IFXE), and normalization of serum free light chains (FLC) and their ratio. However, a follow-up PET-CT revealed new extramedullary plasmacytomas that developed despite the serologic improvement. Unfortunately, her clinical course was further complicated by several pathologic fractures resulting from extensive skeletal involvement, which eventually required surgical repair.
The transition from RVD to second-line therapy was delayed by 6 weeks to allow for fracture healing. Standard-dose carfilzomib, daratumumab, and dexamethasone (DKD) were initiated, resulting in a partial response (PR) by PET-CT and continued normalization of serum light chains. During the third cycle, she developed a normocytic anemia out of proportion to the expected pancytopenia of the regimen. Further workup revealed iron of 15, iron saturation of 6%, and a ferritin of 74. An iron infusion was given; however, the patient’s state of anemia continued, and she became transfusion dependent. Repeat iron studies demonstrated persistent iron deficiency, raising concerns about asymptomatic blood loss, and an urgent endoscopy was planned.
Before endoscopy could be performed, she presented emergently with acute abdominal pain, oral intolerance, and obstipation. A CT of the abdomen demonstrated an enteroenteric intussusception in the left lower quadrant, resulting in proximal small bowel obstruction (SBO). Within the intussusception, there was a suggestion of an intraluminal soft tissue mass measuring up to 2.2 cm (Fig. 3). The general surgery service was consulted for evaluation, and a nasogastric tube was placed. Within 24 h of presentation, the patient was taken to the operating room for a diagnostic laparoscopy after multidisciplinary discussion. Direct visualization revealed extensively dilated loops of proximal small bowel from an intussusception secondary to the intraluminal mass, prompting consultation with surgical oncology. The operation was converted to a vertical infraumbilical laparotomy to allow for a formal oncological resection. The involved segment of small bowel was resected with 5 cm margins proximally and distally to allow for appropriate mesentery and lymph node sampling.
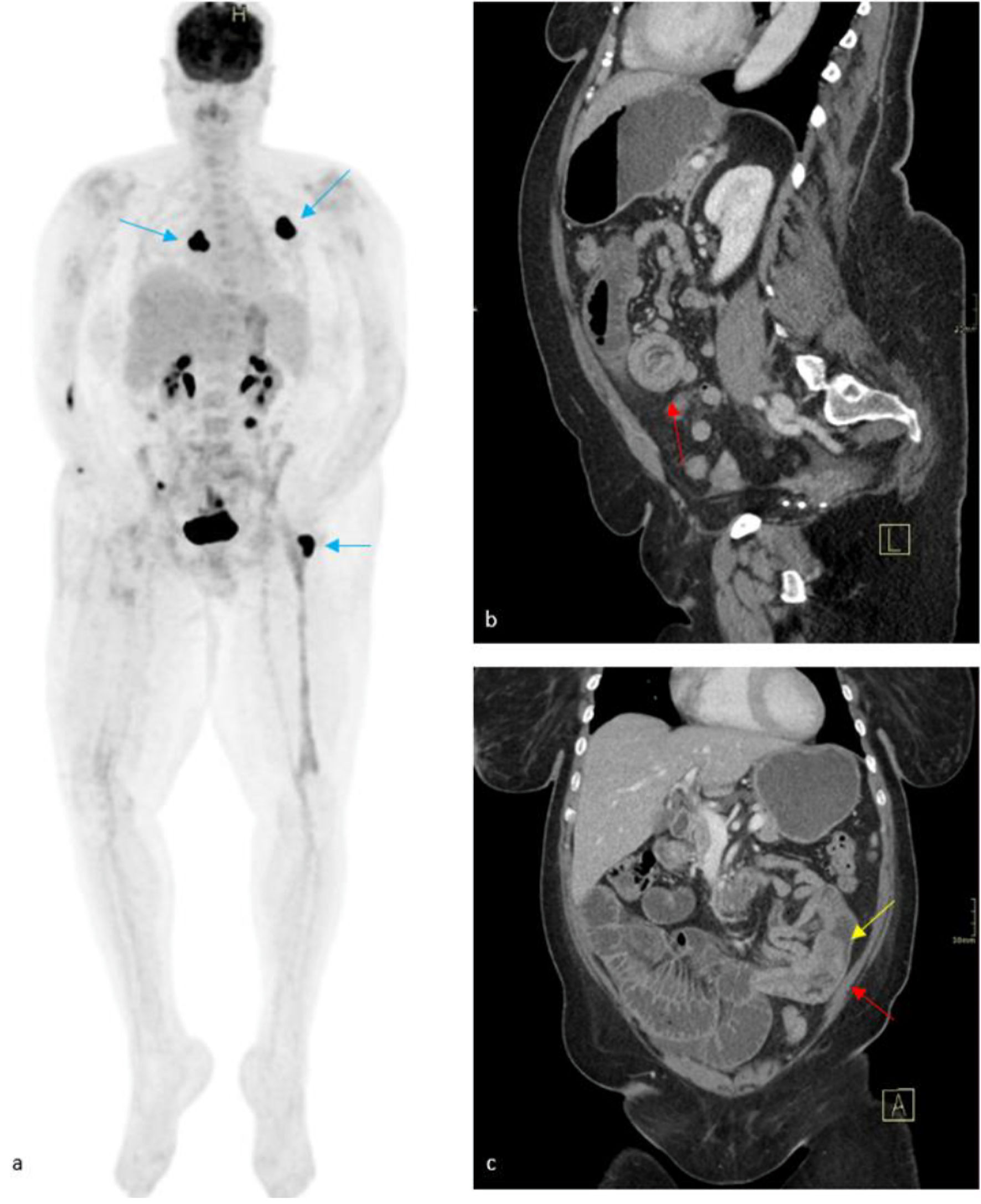 Click for large image | Figure 3. Maximum intensity projection (MIP) FDG PET/CT (a), which was taken 2 months prior to presentation, demonstrated mixed response to medical therapy, with avid radiotracer uptake seen within masses of the left posterior fourth rib, right posterior thoracic soft tissues, and left greater trochanter (blue arrows). Physiologic radiotracer activity is seen within the renal collecting system, urinary bladder, and areas of small bowel. Contrasted CT (b, c) revealed an enteroenteric intussusception (red arrows) in the left lower quadrant with upstream small bowel obstruction. A soft-tissue mass is suspected as the lead point for the intussusception (yellow arrow) with a decompressed distal small bowel. FDG PET/CT: fluorodeoxyglucose positron emission tomography/computed tomography. |
Gross examination of the resection specimen revealed an exophytic intraluminal small bowel mass measuring 2.9 cm in dimension and a second focus measuring 0.5 cm in dimension representing an effaced lymph node. Histologic examination demonstrated diffuse proliferation of atypical plasma cells with a loss of normal lymph node architecture. CD138 immunohistochemistry (IHC) highlighted the neoplastic cells, and Epstein Barr virus (EBV) in situ hybridization was negative. Kappa/lambda IHC showed variable positivity for kappa and was negative for lambda in the neoplastic cells (Fig. 4).
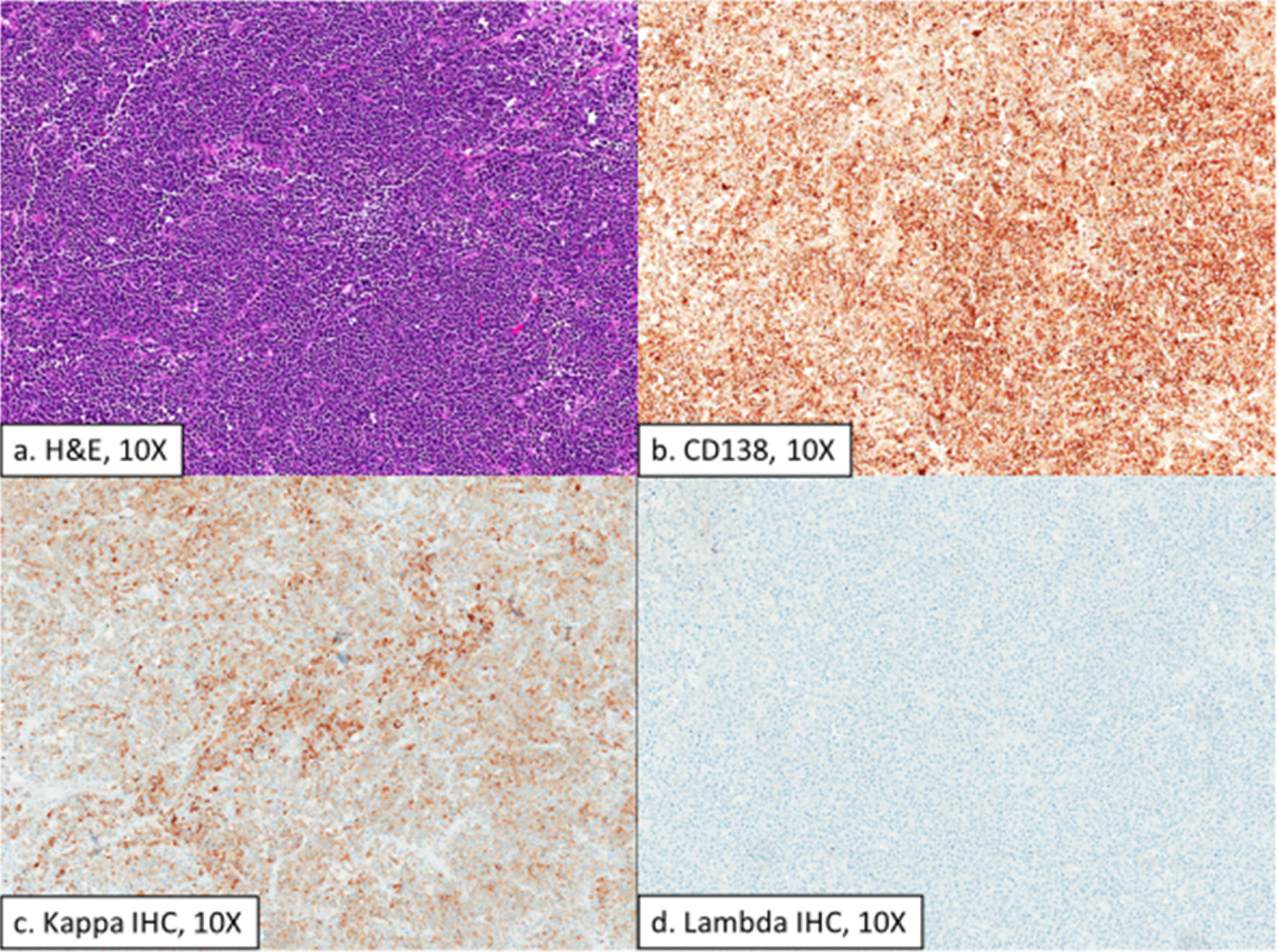 Click for large image | Figure 4. Histologic sections of a large exophytic intraluminal small bowel mass showing a high-grade plasma cell neoplasm (a) that is positive for CD138 (b), variably positive for Kappa IHC (c), and negative for lambda IHC (d). H&E: hematoxylin and eosin; IHC: immunohistochemistry. |
Upon recovery, cytotoxic chemotherapy was initiated with bortezomib, dexamethasone, thalidomide, cisplatin, cyclophosphamide, etoposide (VTD-PCE). Normally, we would have used VTD-PACE (the same regimen but with doxorubicin). However, in this case, doxorubicin was held due to concurrent radiation being delivered for extension of a dural-based plasmacytoma. Two cycles of VTD-PCE induced a VGPR based on PET-CT, and she subsequently underwent consolidation with melphalan followed by auto-SCR. Unfortunately, her clinical course was further complicated by progressive hepatic plasmacytomas, which ultimately led to acute hepatic failure and eventual death while under home hospice care.
| Discussion | ▴Top |
Extramedullary myeloma represents a notably severe form of MM, defined by the existence of the disease beyond the bone marrow. While EMM is increasingly recognized for its complex cytogenetics, refractory disease, and adverse clinical outcomes, there is a paucity of literature addressing specific treatment regimens. One challenge is that there are no strict universal clinical criteria by which to define EMM; however, it is largely defined by myeloma cells that escape the bone marrow and invade tissues [13].
Treatment regimens are based on therapies used for MM; however, the wide range of available treatment modalities presents clinicians with many options. Additionally, the frontier of MM is rapidly evolving with frequent novel therapies emerging. Given that EMM is, at its core, high-risk MM, many of the treatment regimens are extrapolated from data based on treating high-risk MM. Lenalidomide appears to be largely effective [14], but chiefly when combined with a proteasome inhibitor, with bortezomib in particular demonstrating efficacy in EMM [15]. This is a reflection of bortezomib’s propensity to treat high-risk disease, with action on overcoming t(4;14) and del(17p) [16]. Commonly used starting regimens include carfilzomib, lenalidomide, dexamethasone (KRD), and VRD, with KRD showing no progression-free survival (PFS) with more toxicity and treatment-related deaths in a trial, which notably excluded high-risk patients [17]. With this in mind, standard VRD was started up front, which resulted in a VGPR by serum markers despite persistence of plasmacytomas [18-21].
Even though she was in a serologic VGPR, this was not an accurate characterization of her disease status given her persistent plasmacytomas. Hence, the next targeted regimen was aimed at relapsed refractory MM in DKD. The decision to use DKD was based on studies demonstrating efficacy in refractory disease in addition to data supporting the use of carfilzomib for high-risk patients [22]. However, the data in EMM were based primarily on case reports given the paucity of literature supporting specific regimens to treat EMM. Some reports suggest that daratumumab may have limited efficacy in EMM due to decreased levels of CD38 expression by EMM plasma cells [23]. However, one trial demonstrated modest efficacy in EMM [24], which supported our use in this patient. Carfilzomib appears to be effective in patients with high-risk features, but there are a limited number of case reports mentioning its use in extramedullary disease, with one case specifically mentioning a durable complete response when combined with dexamethasone [25].
Despite being placed on both first- and second-line therapies involving these agents, our patient’s disease progressed, as evidenced by the development of new extramedullary plasmacytomas even while her serologic markers indicated a PR. This points to the limitations of relying on serum M-protein or light chains for disease monitoring in EMM. Unlike conventional MM, where these markers provide reliable indications of disease burden, EMM is often characterized by non-secretory or low-secretory behavior, where tumor cells produce minimal or no detectable M-protein or FLC. This discrepancy highlighted one of the unique challenges in managing EMM: traditional serologic markers such as M-protein and serum-FLC can fail to accurately reflect disease burden in extramedullary cases, especially in non-secretory or low-secretory disease [26].
Given her numerous complications and persistent disease, she was initially unable to meet the criteria for auto-SCR, necessitating the use of these therapies to manage her complications with the goal of creating an opportunity for auto-SCR in the future. One of the most difficult aspects of managing EMM is its poor response to medications that make up the cornerstone of most standard MM regimens. In this case, the disease was refractory to first- and second-line therapies with proteasome inhibitors and IMiDs, necessitating the transition to a more intensive cytotoxic chemotherapy regimen with VTD-PACE (modified to VTD-PCE). This regimen is commonly used to bridge auto-SCR, particularly in the subset of patients such as plasma cell leukemia or EMM. The American Society of Clinical Oncology (ASCO) guidelines suggest using it in this population [27], while some studies support using it for severe relapsed or refractory disease [28].
In addition to standard chemotherapy, radiation therapy was a key part of managing this patient’s disease and was aimed chiefly at symptom improvement. Radiation is particularly useful for controlling localized tumor burden and relieving symptoms tied to plasmacytomas [27], such as spinal cord compression and SBO, as seen in this case [29]. However, the use of radiation in EMM must be carefully considered to avoid overlapping toxicities, especially when used alongside systemic treatments. To avoid complications, doxorubicin was held in the VTD-PACE regimen (VTD-PCE) to limit the risk of radiation recall when used in conjunction with radiation. This reinforces the value of a multimodal approach (combining systemic therapy, radiation, and potentially surgery) as a strategy for achieving disease control in patients with EMM.
Other complications of this case were related to treatment toxicities. While pancytopenia is expected with DKD, when anemia occurs out of proportion to the expected degree of neutropenia and thrombocytopenia, assessment for concomitant etiologies for anemia should be considered. In a study comparing DKD to other regimens, the most common grade 3 or higher hematologic adverse events in the DKD group were thrombocytopenia (32%), anemia (22%), and neutropenia (16%) [30]. Another recent analysis of the original study of DKD demonstrated thrombocytopenia as a much more common side effect than anemia, with severe anemia appearing in 17.5% of patients, compared to 24.7% of patients presenting with thrombocytopenia [31]. Thus, the suppression of erythropoiesis (leading to anemia) is typically part of a broader pattern of bone marrow suppression that also affects leukopoiesis and thrombopoiesis, resulting in neutropenia and thrombocytopenia [32].
In this case, the presence of severe iron deficiency anemia prompted assessment for malabsorption and blood loss. Given the steroid backbone for myeloma-directed therapy, peptic ulcer disease was the leading differential diagnosis, despite proton pump inhibitor (PPI) therapy and the absence of gastrointestinal (GI) symptoms. Unexpectedly, in a matter of weeks, she developed an SBO secondary to a plasmacytoma in the setting of normal serum light chains. Here, discussions regarding the benefits of cytotoxic chemotherapy in treating EMM were carefully weighed against potential toxicities. This is paramount in these patients, particularly in cases with compromised organ function or reduced performance status due to the underlying condition. The patient’s widespread disease had already led to pathologic fractures and worsening anemia, adding complexity to her care and causing treatment delays. Combination regimens that include alkylating agents and other cytotoxic drugs require vigilant monitoring for adverse effects, as these therapies can significantly affect blood counts and heighten the risk of infections or organ damage. Despite these risks, aggressive treatment may be warranted in cases of refractory EMM, where survival is often less than 6 months, to improve response rates and extend survival.
The cytogenetic profile seen in EMM is an area of ongoing research. The many genetic abnormalities such as p53 mutations, CCND1/IGH translocations, and multiple chromosomal variations suggest that EMM may have a distinct genomic landscape that drives its aggressive phenotype, leading to its treatment resistance [33]. The t(11; 14), normally not a particularly high-risk translocation, contributes to loss of bone marrow homing machinery and is often seen in plasma-cell leukemia (PCL). The high prevalence of t(11;14) in PCL, which is characterized by the presence of circulating plasma cells, suggests that this translocation may play a role in enabling plasma cells to survive and proliferate outside the bone marrow. Further studies are needed, but this is supported by findings that, on occasion, PCL and extramedullary disease (EMD) share certain cytogenetic features, including t(11;14) [34].
Studies focused on the molecular and genetic characteristics of EMM could yield insights into targeted therapies tailored to these specific mutations, potentially improving outcomes. For example, agents targeting the p53 pathway, checkpoint inhibitors, or novel small molecules that interfere with specific translocations may offer new avenues for treating EMM. Until these targeted approaches become available, cytotoxic chemotherapy and a tailored, multimodal approach remain the mainstay of treatment, although their efficacy is limited. As the frontier of MM continues to evolve, therapies like CAR-T, BiTEs, and monoclonal antibodies are likely to play a larger role in therapy, potentially improving outcomes in high-risk patients such as those with EMM. With up-and-coming therapies, perhaps the need for autologous transplant will become less of a prominent therapy for high-risk MM.
A critical tool that has emerged for the evaluation of extramedullary disease is PET-CT, allowing for the detection of extramedullary plasmacytomas and monitoring treatment response. This patient’s disease is remarkable both for the complex cytogenetics and that her plasmacytomas progressed on standard first- and second-line MM-directed therapy despite normal serum markers. Thus, serial PET-CTs were required to monitor her disease response to therapy, which is not required in standard MM but has become increasingly recognized in the management of EMM [5, 35]. In this patient’s case, her serum markers were unreliable because she continued to develop extramedullary plasmacytomas. The American Society of Hematology (ASH) in its “How I Treat EMM” recommends the use of PET-CT for surveillance in the context of a clinical concern for EMM, light-chain escape, or non-secretory disease [5]. This is supported by the International Myeloma Working Group’s recommendation for the use of PET-CT, in which the modality is utilized more regularly to distinguish between active and inactive disease [36]. PET-CT is not currently included in the National Comprehensive Cancer Network (NCCN) criteria for response assessment in MM, but its role in EMM is gaining traction. This supports the need for consensus guidelines that incorporate PET-CT as a standard tool in EMM, as it can be essential for accurate disease assessment and therapeutic decision-making in this subset of myeloma patients.
Conclusions
Refractory EMM carries a dismal prognosis, with overall survival often less than 6 months. Given the extent of extramedullary disease and central nervous system (CNS) involvement at presentation, earlier aggressive treatment might have prevented complications such as the small bowel plasmacytoma, though baseline functional status limited this option. Delays in systemic therapy are detrimental, emphasizing the critical need for clinical trials to elucidate EMM’s unique pathophysiology and improve outcomes. Subgroup analyses in larger prospective trials could provide insights into tailored treatment approaches for this aggressive subtype.
Imaging with PET-CT enabled accurate assessment of disease progression and treatment response, as traditional serologic markers were unreliable due to the non-secretory nature of the disease. Guidelines containing standardized imaging protocols could enhance clinicians’ ability to monitor disease burden, particularly in non-secretory or low-secretory cases such as these.
Ultimately, this case underscores the need for a highly individualized approach to EMM, leveraging molecular insights, advanced imaging, and multimodal therapies to address its complex manifestations. It highlights the unique treatment challenges, including the limitations of standard myeloma therapies. Advanced imaging monitoring with PET-CT is important, as serologic monitoring may prove unreliable. Finally, the necessity of a multimodal approach was emphasized: radiation therapy effectively controlled localized plasmacytomas, while systemic responses required alkylator-based cytotoxic chemotherapy. By prioritizing clinical trials, molecular profiling, and personalized treatment strategies, the medical community can advance toward better outcomes for patients with this challenging and aggressive form of myeloma.
Acknowledgments
The views expressed herein are those of the author(s) and do not reflect the official policy or position of the Defense Health Agency, Brooke Army Medical Center, the Department of Defense, or any agencies under the US government.
Financial Disclosure
Funding is being applied for through the Geneva Foundation but has not yet been granted.
Conflict of Interest
No party/author has any conflict of interest.
Informed Consent
The patient has signed the informed consent and has read and approved the draft.
Author Contributions
Each author contributed equally to this case report. Paul J. Wurtz and Kevin McGovern wrote components, assisted with image layout and captions and edited the manuscript. Jamie Shah performed literature review, prepared pathology images and caption and edited the manuscript. Devin Moore wrote components of the manuscript and edited the completed manuscript. Kristin E. Stoll performed literature review, wrote components and edited the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Cowan AJ, Green DJ, Kwok M, Lee S, Coffey DG, Holmberg LA, Tuazon S, et al. Diagnosis and management of multiple myeloma: a review. JAMA. 2022;327(5):464-477.
doi pubmed - Zanwar S, Ho M, Lin Y, Kapoor P, Binder M, Buadi FK, Dispenzieri A, et al. Natural history, predictors of development of extramedullary disease, and treatment outcomes for patients with extramedullary multiple myeloma. Am J Hematol. 2023;98(10):1540-1549.
doi pubmed - Blade J, Fernandez de Larrea C, Rosinol L, Cibeira MT, Jimenez R, Powles R. Soft-tissue plasmacytomas in multiple myeloma: incidence, mechanisms of extramedullary spread, and treatment approach. J Clin Oncol. 2011;29(28):3805-3812.
doi pubmed - Usmani SZ, Heuck C, Mitchell A, Szymonifka J, Nair B, Hoering A, Alsayed Y, et al. Extramedullary disease portends poor prognosis in multiple myeloma and is over-represented in high-risk disease even in the era of novel agents. Haematologica. 2012;97(11):1761-1767.
doi pubmed - Touzeau C, Moreau P. How I treat extramedullary myeloma. Blood. 2016;127(8):971-976.
doi pubmed - Weinstock M, Ghobrial IM. Extramedullary multiple myeloma. Leuk Lymphoma. 2013;54(6):1135-1141.
doi pubmed - Liu Y, Jelloul F, Zhang Y, Bhavsar T, Ho C, Rao M, Lewis NE, et al. Genetic basis of extramedullary plasmablastic transformation of multiple myeloma. Am J Surg Pathol. 2020;44(6):838-848.
doi pubmed - McAvera R, Quinn J, Murphy P, Glavey S. Genetic abnormalities in extramedullary multiple myeloma. Int J Mol Sci. 2023;24(14):11259.
doi pubmed - Weinstock M, Aljawai Y, Morgan EA, Laubach J, Gannon M, Roccaro AM, Varga C, et al. Incidence and clinical features of extramedullary multiple myeloma in patients who underwent stem cell transplantation. Br J Haematol. 2015;169(6):851-858.
doi pubmed - Varettoni M, Corso A, Pica G, Mangiacavalli S, Pascutto C, Lazzarino M. Incidence, presenting features and outcome of extramedullary disease in multiple myeloma: a longitudinal study on 1003 consecutive patients. Ann Oncol. 2010;21(2):325-330.
doi pubmed - Beksac M, Tuglular T, Gay F, Mina R, Katodritou E, Unal A, Cavo M, et al. Efficacy of daratumumab combined with bortezomib, cyclophosphamide and dexamethasone for the treatment of multiple myeloma patients presenting with extramedullary disease: A European Myeloma Network study (EMN19). Blood. 2022;21(Suppl 1):10177-10179.
doi - Rosinol L, Beksac M, Zamagni E, Van de Donk N, Anderson KC, Badros A, Caers J, et al. Expert review on soft-tissue plasmacytomas in multiple myeloma: definition, disease assessment and treatment considerations. Br J Haematol. 2021;194(3):496-507.
doi pubmed - Chen Y, Tao S, Zheng X, Shi Y, Zhang L, Chen K, He Z, et al. Research progress on treatment of extramedullary multiple myeloma. Hematology. 2021;26(1):985-994.
doi pubmed - Wang J, Shen N, Shen X, Zhang R, Jin Y, Li J, Chen L. Survival trends and prognostic factors of patients with newly diagnosed multiple myeloma accompanied with extramedullary disease. Ann Med. 2023;55(2):2281657.
doi pubmed - Laura R, Cibeira MT, Uriburu C, Yantorno S, Salamero O, Blade J, Montserrat E. Bortezomib: an effective agent in extramedullary disease in multiple myeloma. Eur J Haematol. 2006;76(5):405-408.
doi pubmed - El-Ghammaz AM, Abdelwahed E. Bortezomib-based induction improves progression-free survival of myeloma patients harboring 17p deletion and/or t(4;14) and overcomes their adverse prognosis. Ann Hematol. 2016;95(8):1315-1321.
doi pubmed - Kumar SK, Jacobus SJ, Cohen AD, Weiss M, Callander N, Singh AK, Parker TL, et al. Carfilzomib or bortezomib in combination with lenalidomide and dexamethasone for patients with newly diagnosed multiple myeloma without intention for immediate autologous stem-cell transplantation (ENDURANCE): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2020;21(10):1317-1330.
doi pubmed - Voorhees PM, Sborov DW, Laubach J, Kaufman JL, Reeves B, Rodriguez C, Chari A, et al. Addition of daratumumab to lenalidomide, bortezomib, and dexamethasone for transplantation-eligible patients with newly diagnosed multiple myeloma (GRIFFIN): final analysis of an open-label, randomised, phase 2 trial. Lancet Haematol. 2023;10(10):e825-e837.
doi pubmed - Moreau P, Hulin C, Perrot A, Arnulf B, Belhadj K, Benboubker L, Bene MC, et al. Maintenance with daratumumab or observation following treatment with bortezomib, thalidomide, and dexamethasone with or without daratumumab and autologous stem-cell transplant in patients with newly diagnosed multiple myeloma (CASSIOPEIA): an open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22(10):1378-1390.
doi pubmed - Facon T, Kumar SK, Plesner T, Orlowski RZ, Moreau P, Bahlis N, Basu S, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
doi pubmed - Mateos MV, Dimopoulos MA, Cavo M, Suzuki K, Jakubowiak A, Knop S, Doyen C, et al. Daratumumab plus bortezomib, melphalan, and prednisone for untreated myeloma. N Engl J Med. 2018;378(6):518-528.
doi pubmed - Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, Weisel K, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): updated outcomes from a randomised, multicentre, open-label, phase 3 study. Lancet Oncol. 2022;23(1):65-76.
doi pubmed - Byun JM, Min CK, Kim K, Bang SM, Lee JJ, Kim JS, Yoon SS, et al. Phase II trial of daratumumab with DCEP in relapsed/refractory multiple myeloma patients with extramedullary disease. J Hematol Oncol. 2022;15(1):150.
doi pubmed - Lonial S, Weiss BM, Usmani SZ, Singhal S, Chari A, Bahlis NJ, Belch A, et al. Daratumumab monotherapy in patients with treatment-refractory multiple myeloma (SIRIUS): an open-label, randomised, phase 2 trial. Lancet. 2016;387(10027):1551-1560.
doi pubmed - Espanol I, Romera M, Gutierrez-Meca MD, Garcia MDC, Tejedor A, Martinez A, Ibanez J, et al. Carfilzomib and dexamethasone for extramedullary myeloma with pleuropericardial involvement. Clin Case Rep. 2017;5(8):1258-1260.
doi pubmed - Drayson M, Tang LX, Drew R, Mead GP, Carr-Smith H, Bradwell AR. Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood. 2001;97(9):2900-2902.
doi pubmed - Mikhael J, Ismaila N, Cheung MC, Costello C, Dhodapkar MV, Kumar S, Lacy M, et al. Treatment of multiple myeloma: ASCO and CCO joint clinical practice guideline. J Clin Oncol. 2019;37(14):1228-1263.
doi pubmed - Togano T, Andoh S, Komuro M, Mitsui Y, Itoi S, Hirai R, Nakamura M, et al. Bortezomib-thalidomide-dexamethasone-cisplatin-doxorubicin-cyclophosphamide-etoposide as a Salvage and Bridging Regimen before Hematopoietic Stem Cell Transplantation for Relapsed or Refractory Multiple Myeloma. Intern Med. 2022;61(22):3329-3334.
doi pubmed - Talamo G, Dimaio C, Abbi KK, Pandey MK, Malysz J, Creer MH, Zhu J, et al. Current role of radiation therapy for multiple myeloma. Front Oncol. 2015;5:40.
doi pubmed - Weisel K, Dimopoulos MA, Beksac M, Leleu X, Richter J, Heeg B, Patel S, et al. Carfilzomib, daratumumab, and dexamethasone (KdD) vs. lenalidomide-sparing pomalidomide-containing triplet regimens for relapsed/refractory multiple myeloma: an indirect treatment comparison. Leuk Lymphoma. 2024;65(4):481-492.
doi pubmed - Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, Weisel K, et al. Final analysis of carfilzomib, dexamethasone, and daratumumab vs carfilzomib and dexamethasone in the CANDOR study. Blood Adv. 2023;7(14):3739-3748.
doi pubmed - Byun JM, Park SS, Yoon SS, Ahn A, Kim M, Lee JY, Jeon YW, et al. Advantage of achieving deep response following frontline daratumumab-VTd compared to VRd in transplant-eligible multiple myeloma: multicenter study. Blood Res. 2023;58(2):83-90.
doi pubmed - Bustoros M, Mejia Saldarriaga M, Jayabalan D, et al. Genomic landscape of multiple myeloma with extramedullary disease: Results from a large patient database. J Clin Oncol. 2023;41(16_suppl):8058.
doi - Cazaubiel T, Leleu X, Perrot A, Manier S, Buisson L, Maheo S, Do Souto Ferreira L, et al. Primary plasma cell leukemias displaying t(11;14) have specific genomic, transcriptional, and clinical features. Blood. 2022;139(17):2666-2672.
doi pubmed - Walker RC, Brown TL, Jones-Jackson LB, De Blanche L, Bartel T. Imaging of multiple myeloma and related plasma cell dyscrasias. J Nucl Med. 2012;53(7):1091-1101.
doi pubmed - Cavo M, Terpos E, Nanni C, Moreau P, Lentzsch S, Zweegman S, Hillengass J, et al. Role of (18)F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: a consensus statement by the International Myeloma Working Group. Lancet Oncol. 2017;18(4):e206-e217.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.