| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Case Report
Volume 000, Number 000, January 2025, pages 000-000
Combined Impact of Prior Polatuzumab Vedotin Plus Bendamustine and Rituximab Therapy and Myeloablative Conditioning on Early Post-Transplant BK Virus-Associated Hemorrhagic Cystitis
Yudai Hayashia , Masao Tsukadaa, b, Daisuke Shinodaa, Marina Matsuia, Kanichi Iwamaa, Koichi Kajiwaraa, Yasuji Kozaia
aDepartment of Hematology, Tokyo Metropolitan Tama Medical Center, Fuchu City, Tokyo, Japan
bCorresponding Author: Masao Tsukada, Department of Hematology, Tokyo Metropolitan Tama Medical Center, Fuchu City, Tokyo 183-8524, Japan
Manuscript submitted December 9, 2024, accepted January 18, 2025, published online January xx, 2025
Short title: Impact of Pola-BR on BKV-HC
doi: https://doi.org/10.14740/jh2010
| Abstract | ▴Top |
Relapsed/refractory diffuse large B-cell lymphomas (R/R DLBCLs) have an extremely poor prognosis, with no established salvage chemotherapy currently available. Polatuzumab, rituximab, and bendamustine combination therapy (Pola-BR) has been approved as a new therapeutic option for R/R DLBCL. Recently, chimeric antigen receptor T-cell therapy and bispecific antibodies have induced long-term remission in many patients with R/R DLBCL. However, allogeneic transplantation remains potentially curative for patients unresponsive to the abovementioned treatments. While allogeneic transplantation can also cause various adverse events, hemorrhagic cystitis is a particularly severe complication that requires effective prevention strategies. Here, we report two cases of severe BK virus-associated hemorrhagic cystitis (BKV-HC) that developed after successive cord blood transplantation with myeloablative conditioning and Pola-BR treatment for early-relapsed DLBCL. Both patients received Pola-BR after undergoing multiple salvage therapies and developed early-onset BKV-HC post-transplant, demonstrating the effects of Pola-BR and myeloablative conditioning. We analyzed the shared characteristics between these two cases to distinguish between the factors that trigger the onset of BKV-HC and those that contribute to its severity. Based on the differences in the clinical course between the two cases, we propose prevention strategies for BKV-HC and identify treatment strategies for Pola-BR in patients with R/R DLBCL undergoing allogeneic transplantation.
Keywords: BK virus; Hemorrhagic cystitis; Allogeneic transplantation; Pola-BR; Polatuzumab; Relapsed/refractory diffuse large B-cell lymphoma
| Introduction | ▴Top |
Polatuzumab vedotin in combination with rituximab, cyclophosphamide, doxorubicin, and prednisone (Pola-R-CHP) has been proven effective for initial diffuse large B-cell lymphoma (DLBCL) treatment and is now considered standard therapy for DLBCL [1]. However, treatment-resistant and early-relapsed DLBCLs have an extremely poor prognosis [2]. Several salvage chemotherapy regimens for relapsed/refractory DLBCL (R/R DLBCL) have been established, including polatuzumab-rituximab-bendamustine combination therapy (Pola-BR) [3], rituximab plus gemcitabine, dexamethasone, and cisplatin (R-GDP), cyclophosphamide, cytarabine, etoposide, dexamethasone, and rituximab (CHASER), and rituximab, ifosfamide, carboplatin and etoposide (R-ICE). However, no effective chemotherapeutic regimen has been established [4]. Although outcomes have improved with the advent of chimeric antigen receptor (CAR) T-cell therapy [5-7] and bispecific antibodies, such as epcoritamab [8], allogeneic transplantation remains a potentially curative option for DLBCL with inadequate or poor treatment response. Achieving deep remission prior to allogeneic transplantation is crucial for improving outcomes. However, allogeneic transplantation can cause various adverse events, such as graft-versus-host disease (GVHD), and has a high non-relapse mortality rate [9, 10]. Hemorrhagic cystitis (HC) is a severe complication of allogeneic transplantation that causes decreased activities of daily living, prolonged hospitalization, and even fatal complications. HC can be either early-onset HC, which is caused by direct bladder mucosal damage from conditioning regimens, or late-onset HC, which is primarily due to viral causes. Viruses that commonly cause HC include adenovirus, BK virus, and JC virus, with BK virus-associated hemorrhagic cystitis (BKV-HC) occurring in 7-54% of adult transplant recipients [11]. Symptoms include microscopic hematuria, with some cases requiring bladder irrigation, and more severe cases can cause decreased renal function, reduced overall survival, and increased treatment-related mortality [12, 13]. No evidence-based treatments are currently available, making the prevention of onset and progression of BKV-HC an important challenge [14].
Here, we report two cases of early-onset BKV-HC following allogeneic transplantation after Pola-BR therapy for R/R DLBCL. Few cases treated with Pola-BR before allogeneic transplantation have been reported, and the impact of this chemotherapeutic regimen on transplantation remains unknown. In the present cases, the development of early-onset BKV-HC after Pola-BR therapy indicates its potential influence. To the best of our knowledge, no reports of BKV-HC following Pola-BR therapy have been published. We analyzed common factors between the two cases, which included the implementation of cord blood transplantation with myeloablative conditioning, cytomegalovirus (CMV) infection, and acute GVHD (aGVHD), to distinguish between factors triggering BKV-HC onset and those contributing to its severity. Based on the differences in the clinical course between the cases, we subsequently propose prevention strategies for both the development and progression of BKV-HC, as well as treatment strategies for Pola-BR administration prior to allogeneic transplantation.
| Case Reports | ▴Top |
Case 1
A 49-year-old Asian woman presented with lower abdominal pain in May 2020 and ascites in July 2020. Ultrasound examination revealed enlargement of the greater omentum and ovaries. In September, omental biopsy confirmed germinal center B-cell like DLBCL cStage IV (intra-abdominal mass, ovarian involvement, and ascites). Rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) therapy was initiated on September 15, 2020. Positron emission tomography/computed tomography (PET/CT) conducted after six treatment cycles on February 28, 2021, revealed progressive metabolic disease (PMD) with new uptake in the perihepatic and perisplenic regions, left subdiaphragmatic area, left paracolic gutter, and intestinal wall. Second-line CHASER was started on April 1, 2021, but a computed tomography (CT) scan after one cycle showed progressive disease (PD). Third-line R-GDP was administered for two cycles from April 21; however, PET/CT revealed PMD. Fourth-line Pola-BR was initiated on June 7. Because of the continuous increase in lactate dehydrogenase (LDH) during treatment, the disease was assessed as PD, and the patient was scheduled for allogeneic transplantation (CAR-T cell therapy was not covered by the Japanese National Insurance System at that time). The planned graft source was umbilical cord blood. The human lymphocyte antigen (HLA) typing showed two locus mismatches: one at locus A and one at locus B with 6/8 HLA match. The total nucleated cell count and CD34+ cell count were 1.69 × 107/kg and 0.74 × 105/kg, respectively.
Total abdominal radiation therapy (20 Grays) was administered from June 17 to July 1. CT performed on July 15 showed a partial response (PR). Rituximab, cladribine, mitoxantrone, and dexamethasone (R-CMD) therapy was initiated on July 21, followed by myeloablative conditioning (fludarabine 30 mg/m2 + busulfan 3.2 mg/kg + cyclophosphamide 60 mg/kg) during the bone marrow suppression period. Subsequently, the patient experienced fever and epigastric pain and was diagnosed with CMV gastroenteritis on August 2 based on C7-HRP positivity (520/50,000 cells). Treatment with ganciclovir 10 mg/kg was started on August 4, which improved symptoms. Letermovir 240 mg/day was initiated for prophylaxis on August 12, followed by cord blood infusion later on the same day.
Gross hematuria appeared on day 4, and BKV-HC was diagnosed based on BKV positivity (urinary BKV DNA quantification on day 7 was 5.0 × 107 copies/mL). Engraftment was observed on day 19. Although urinary BKV DNA levels decreased by day 27 (2.8 × 107 copies/mL, BKV negative in blood), BKV-HC worsened following the onset of aGVHD grade II (skin 3, gut 0, liver 1) on day 29. Cidofovir (CDV) 1 mg/kg/day was administered on the same day three times per week. BKV-HC symptoms were alleviated with the improvement in aGVHD, and CT on day 38 showed improvement in the left renal pelvis, ureter dilation, and bladder wall thickening. However, diuretic-resistant weight gain emerged, prompting hydrocortisone treatment (50 mg twice daily) for suspected aGVHD. The patient subsequently developed jaundice (total bilirubin (T-bil) 2.6 mg/dL), > 5% weight gain, ascites, and upper abdominal pain, all of which met the diagnostic criteria for classical sinusoidal obstruction syndrome (SOS). Defibrotide was initiated on day 41 for late-onset SOS. On day 44, aGVHD worsened to grade III (skin 3, gut 1, liver 2) concurrent with SOS, thereby requiring first-line treatment with prednisolone (PSL) 1 mg/kg/day. CDV was discontinued on day 46 after adequate urine output was achieved with the improvement in aGVHD. Urinary BKV DNA on day 48 was 2.3 × 107 copies/mL (blood BKV DNA 2.9 × 103 copies/mL).
PSL treatment improved aGVHD, and cyclosporine was gradually discontinued. However, increasing T-bil levels indicated recurrent aGVHD. During this period, lymphocyte counts were low, with decreased absolute numbers and proportions of CD4+ and CD8+ T cells (Fig. 1a , b). Cyclosporine treatment was resumed on day 62, and second-line treatment with allogeneic bone marrow-derived mesenchymal stem cells was initiated (days 63, 67, 71, and 74). However, because of insufficient therapeutic response, the PSL dose could not be reduced, and BKV-HC worsened (day 64: urinary BKV DNA quantification 5.0 × 107 copies/mL; blood BKV DNA 2.9 × 103 copies/mL).
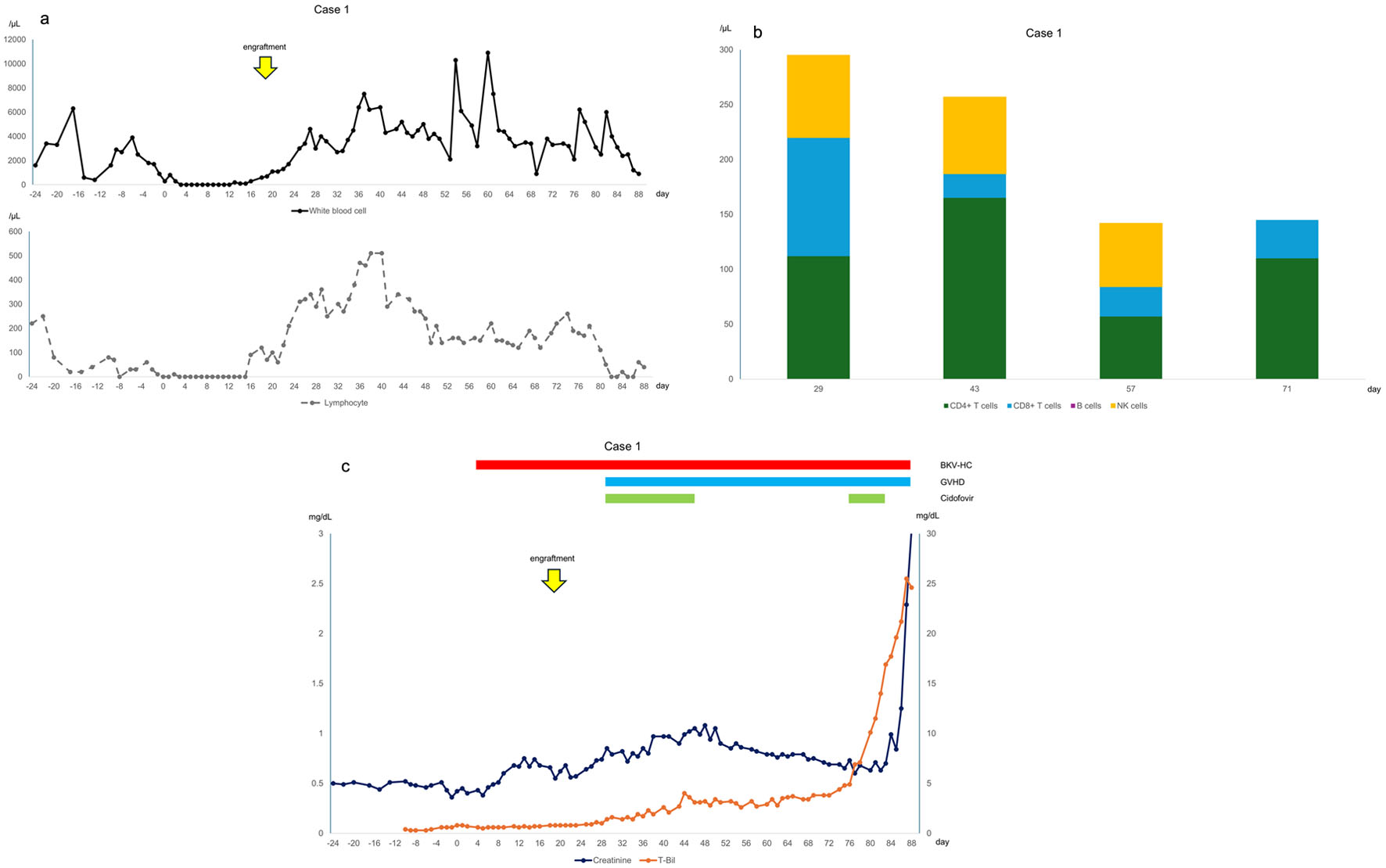 Click for large image | Figure 1. (a) Changes in white blood cell and lymphocyte counts, with day 0 representing the day of infusion. Lymphocyte counts decreased after day 50 when acute graft-versus-host disease (GVHD) worsened again. (b) Lymphocyte subsets, where CD4+ T cells are CD3+CD4+CD8, CD8+ T cells are CD3+CD4CD8+, B cells are CD19+CD20+, and natural killer cells are CD3CD16+CD56+/CD3CD16CD56+. CD4+ T cells were particularly low on day 57 when acute GVHD worsened. (c) Clinical course of the case, with day 0 indicating the day of infusion. BKV-HC: BK virus-associated hemorrhagic cystitis. |
On day 74, the patient was diagnosed with thrombotic microangiopathy based on increased predominantly direct bilirubin, hypertension, increased blood urea nitrogen, increased reticulocytes, presence of schistocytes (2.3%), transfusion-refractory thrombocytopenia, and decreased haptoglobin. Subsequently, cyclosporine administration was discontinued. CDV was restarted to treat BKV-HC on day 76, and steroid therapy was escalated to methylprednisolone (mPSL) at 2 mg/kg/day on day 77 to address worsening aGVHD. Although the mPSL dose was increased, T-bil and LDH continued increasing, prompting mPSL pulse therapy (1 g/day), which was administered on days 82 - 85. Nevertheless, BKV-HC continued to worsen. Although the LDH level stabilized following steroid pulse therapy, it increased to 1,169 U/mL on day 87, indicating DLBCL relapse based on the clinical course. Subsequently, ascites increased and urine output decreased because of deteriorating renal function, leading to death on day 89 (Fig. 1c).
Case 2
A 41-year-old Asian man was diagnosed with Epstein-Barr virus (EBV)-positive DLBCL stage IV (lymph nodes above and below the diaphragm, peritoneum, bone, and spleen) in November 2020. He underwent two cycles of R-CHOP from December. A CT scan performed after the second cycle showed stable disease. Second-line R-GDP was initiated in February 2021. CT performed after two cycles of R-GDP revealed PR. When the patient tested negative for EBV DNA, R-GDP was continued for four cycles. PET/CT in June 2021 showed complete metabolic response (CMR).
In June 2022, elevated serum soluble interleukin-2 receptor (sIL-2R) levels indicated disease recurrence. Because of EBV-positive DLBCL, the patient was ineligible for CAR-T cell therapy and was thus initially planned to undergo autologous transplantation. Third-line CHASER was initiated in November, with CT showing PR after two cycles. Peripheral blood stem cell harvest (PBSCH) was performed in February 2023 after the third CHASER cycle; however, the disease progressed during the fourth cycle (sIL-2R 43,886 U/mL). Because of lymphoma infiltration, as confirmed by bone marrow biopsy, and potential tumor contamination of the PBSCH product, the treatment plan was changed to allogeneic transplantation. The planned graft source was umbilical cord blood. The HLA typing showed two locus mismatches: one at locus A and one at locus DR with 6/8 HLA match. The total nucleated cell count and CD34+ cell count were 1.85 × 107/kg and 0.91 × 105/kg, respectively. Fourth-line Pola-BR was initiated in April. After confirming CMR by PET/CT following the third cycle, four cycles of Pola-BR were completed.
Following myeloablative conditioning with total body irradiation (12 Grays) + cytarabine 3 g/m2 + cyclophosphamide 60 mg/kg starting on June 27, cord blood was infused on July 6. On day 5, urinalysis showed occult blood positivity, and subsequent positive urinary BKV led to the diagnosis of BKV-HC. Gross hematuria worsened on day 21, and CDV was initiated at 1 mg/kg/day three times per week on day 25. Despite deteriorating renal function following initiation, CDV treatment was continued. Engraftment occurred on day 33, and CDV was discontinued on day 34 because of elevated creatinine levels (1.79 mg/dL). Subsequently, renal function improved. Diuretic-resistant weight gain was observed on day 40, accompanied by worsening BKV-HC. Hydrocortisone (100 mg/day) was administered for aGVHD on day 47, and CDV for BKV-HC was resumed on day 54. During this period, lymphocyte counts were low, with decreased absolute numbers and proportions of CD4+ and CD8+ T cells (Fig. 2a, b). On day 59, renal function and BKV-HC worsened, which required increasing the hydrocortisone dosage to 100 mg twice daily for aGVHD and the initiation of bladder irrigation. However, renal function continued to deteriorate, and the patient died of BKV-HC complications on day 73 (Figs. 2c and 3a, b).
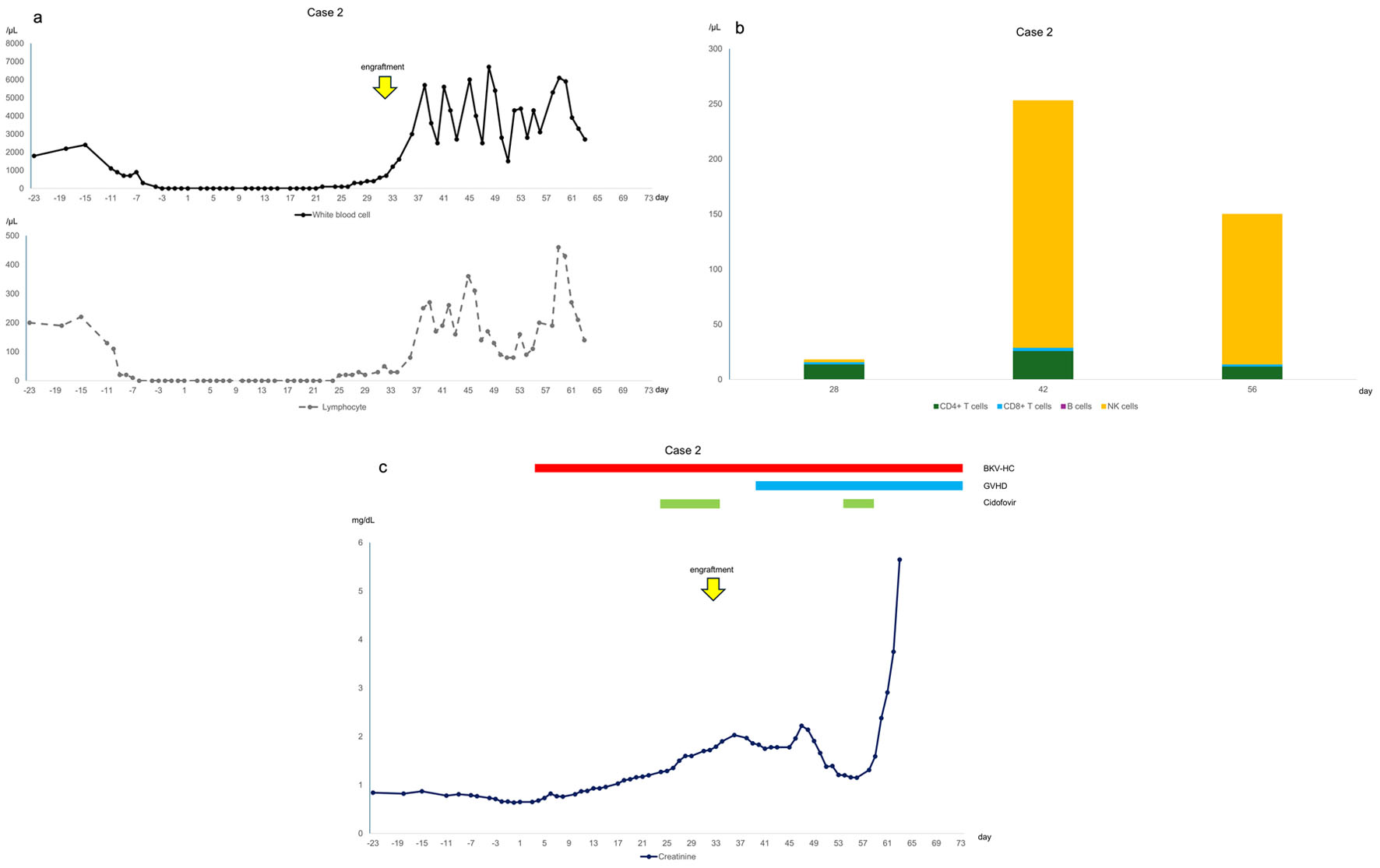 Click for large image | Figure 2. (a) Changes in white blood cell and lymphocyte counts, with day 0 representing the day of infusion. Lymphocyte counts were lower than those in case 1. (b) Lymphocyte subsets. The absolute numbers of CD4+ T cells and CD8+ T cells were lower than those in case 1. (c) Clinical course of the case, with day 0 representing the day of infusion. Creatinine levels increased with the worsening of acute GVHD on day 40. |
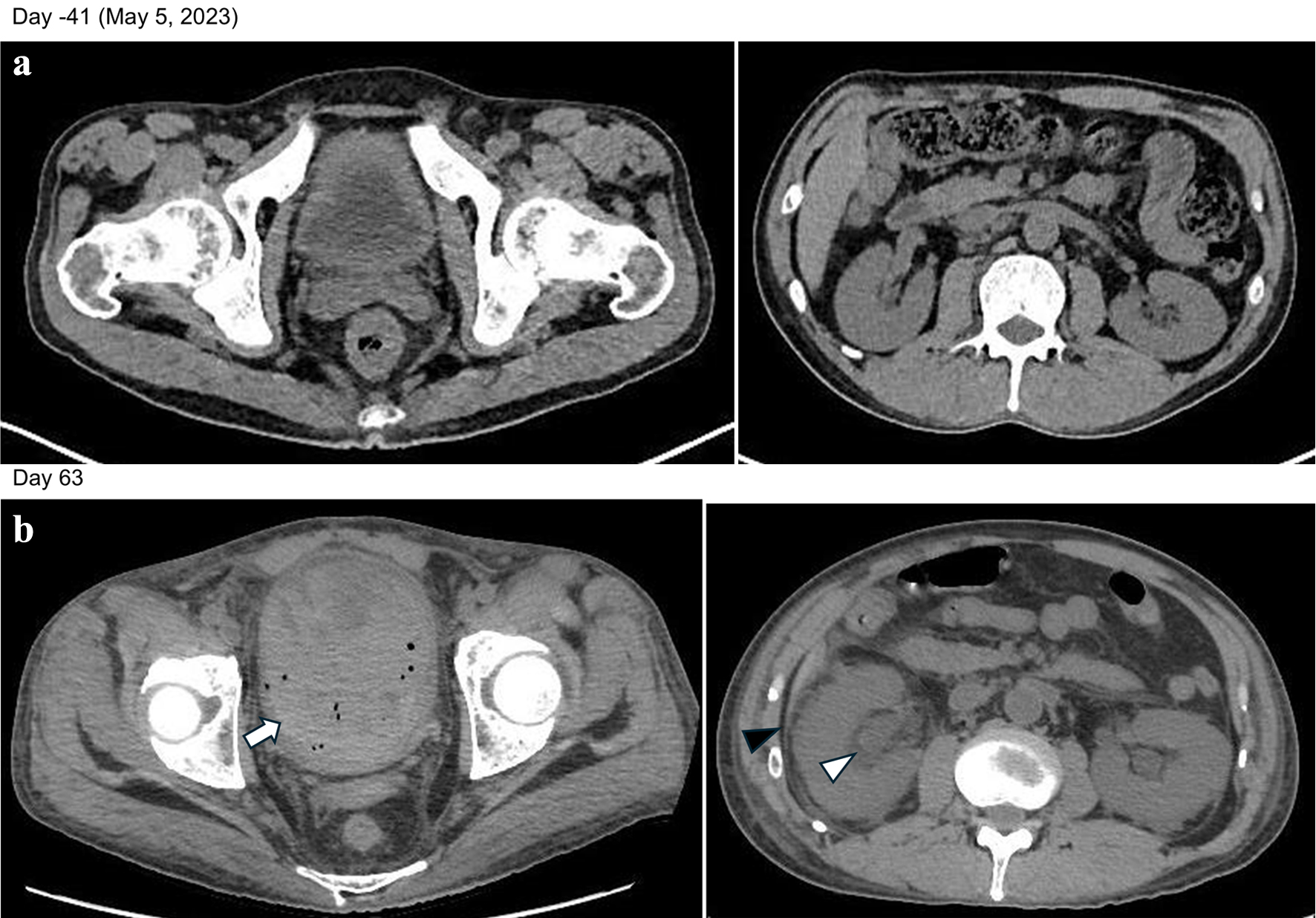 Click for large image | Figure 3. (a) Findings after the completion of three courses of Pola-BR. No abnormal findings were observed in the kidneys. (b) Heterogeneous high-intensity areas in the bladder, indicating hemorrhage and blood clots (arrow). Dilation of the renal pelvis (white arrowhead) and increased density of perirenal fat tissue (black arrowhead) were observed. These findings indicate that the BK virus infection had spread from the bladder to the kidneys. |
| Discussion | ▴Top |
We encountered two cases of severe BKV-HC following allogeneic transplantation after successive salvage therapy that included Pola-BR for R/R DLBCL. Both cases developed BKV-HC in the early post-transplant period. Based on their common features, we categorized the factors into triggering factors (Pola-BR and myeloablative conditioning) and severity factors (cord blood transplantation, CMV infection, and aGVHD). Among the triggering factors, bendamustine is a cytotoxic combination drug that exhibits alkylating and purine analog activities [15]. It delays the recovery of T-cell numbers, particularly CD4+ T cells [16], increasing the risk of viral infections, with late-line use carrying a higher risk [17]. Both cases underwent Pola-BR as fourth-line therapy. Because non-T-cell purging is a known risk factor for BKV-HC [18], polatuzumab may have contributed to the development of BKV-HC. However, no reports have been published on the association between BKV-HC and Pola-BR. Therefore, we suspect that myeloablative conditioning further contributed to BKV-HC development. Myeloablative conditioning is a known risk factor for BKV-HC [19-24] because it causes high-dose drug-induced mucosal damage and prolongs time to engraftment due to intense immunosuppression. The conditioning intensity increases the risk of early (pre-engraftment) BKV-HC. The combination of Pola-BR and myeloablative conditioning likely triggered early BKV reactivation and subsequent HC. Cord blood transplantation, CMV infection, and aGVHD were identified as severity factors. While meta-analysis demonstrated that umbilical cord blood stem cell transplantation did not affect the incidence of BKV-HC [24], some studies have reported that cord blood transplantation carries a higher risk of viral infections than other stem cell sources and increases BKV reactivation risk [13, 25]. A longer time to engraftment can worsen BKV-HC if it develops pre-engraftment. Cord blood transplantation is a high-risk factor for CMV infection due to CMV-negative cord blood. The association between CMV and BKV-HC is well documented [22, 23, 26], with CMV being a possible contributor to BKV DNA replication [27]. Both cases showed persistent CMV antigenemia post-engraftment, potentially contributing to BKV-HC severity. aGVHD is also a known risk factor for BKV-HC [22, 24]. Viral infections themselves can worsen aGVHD and potentially worsen BKV-HC severity. In both cases, BKV-HC worsened during GVHD onset, resulting in renal dysfunction.
The two cases differed significantly in disease progression. Although case 2 maintained a complete response (CR) to treatment, his BKV-HC was more severe than that in case 1, which resulted in death. CD4+ T-cell recovery in cord blood transplantation is slower than in other scenarios, potentially affecting both the quantitative recovery and functional maturation of CD8+ T cells. Case 2 showed lower white blood cell and lymphocyte counts than case 1, with notably marked decreases in both CD4+ and CD8+ T cells. In case 2, the reduction in CD4+ T cells likely diminished the CD8+ T-cell responsiveness against BKV, potentially leading to more severe BKV-HC [28]. Furthermore, the decrease in absolute CD4+ T-cell counts may have caused a relative reduction in regulatory T cells, indicating that worsening aGVHD further exacerbated BKV-HC severity. Certainly, when using Pola-BR before transplantation and increasing the intensity of the conditioning regimen, various viruses including herpes simplex virus, varicella-zoster virus, and CMV may tend to reactivate. However, unlike CMV, for which preventive medication is available, BKV lacks prophylactic options and is reactivated early, leading to HC. Early BKV-HC may further exacerbate other post-transplant complications. Through our cases, we believe it is important to consider the impact of Pola-BR on BKV-HC in allogeneic transplantation. Given the observational nature of this study, there remains uncertainty regarding risk and causality. Further investigation, particularly through a retrospective case-control study, is warranted to establish whether polatuzumab treatment is causally associated with an increased risk of severe HC.
Based on the findings, we propose a new treatment strategy for preventing severe BKV-HC, as observed in case 2. Regarding triggering factors, while Pola-BR increased the risk of BKV-HC, replacing it was not feasible since it had effectively achieved CR. However, considering the conditioning intensity, since CR was achieved, applying reduced-intensity conditioning instead of myeloablative conditioning could have lowered the risk and potentially prevented early post-transplant BKV-HC. Of the severity factors, cord blood transplantation should have been avoided as the stem cell source, particularly considering the risk of CMV coinfection. While bone marrow bank donors would be preferable alternative sources because of earlier engraftment and lower viral infection risk, the waiting period reduces their feasibility, given the disease progression of R/R DLBCL. Furthermore, the higher risk of exacerbating BKV-HC severity posed by aGVHD than that by cord blood transplantation needs to be considered. Therefore, if time constraints preclude waiting for a bone marrow donor, haploidentical transplantation might be a feasible alternative, given its rapid engraftment and lower aGVHD risk and despite its known risk of BKV reactivation [24].
Even with the diversity of treatment options in the current era, including bispecific antibody therapy and CAR-T cell therapy, allogeneic transplantation remains a viable therapeutic option for R/R DLBCL. Although treatments for post-transplant BKV-HC exist, such as CDV [29], no standardized treatment protocol has been established [14]; thus, prevention of BKCV-HC occurrence and severity is crucial. In particular, early post-transplant BKV-HC development, combined with the presence of severity factors, is expected to worsen prognosis. Our analysis of the triggering and severity factors in these cases may facilitate the development of effective strategic approaches for Pola-BR administration in allogeneic transplantation for R/R DLBCL.
Conclusion
Successive Pola-BR therapy and myeloablative conditioning for cord blood transplantation for R/R DLBCL can trigger early post-transplant BKV reactivation and cause severe HC. By analyzing these cases, we identified the factors that triggered BKV-HC and those that contributed to its severity. Thus, this case report demonstrates the importance of reducing both the occurrence and severity of BKV-HC while identifying new treatment strategies for R/R DLBCL.
Learning points
Pola-BR and myeloablative conditioning regimens pose significant risk factors for the development of early post-transplant BKV-HC.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors have no conflict of interest to declare.
Informed Consent
The patients provided written consent for publication.
Author Contributions
Conceptualization: Masao Tsukada. Project administration: Yudai Hayashi, Marina Matsui, and Masao Tsukada. Resources: Yudai Hayashi, Marina Matsui, Masao Tsukada, Daisuke Shinoda, Kanichi Iwama, Koichi Kajiwara, and Yasuji Kozai. Supervision: Yasuji Kozai. Writing - original draft: Yudai Hayashi. All authors reviewed and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
aGVHD: acute graft-versus-host disease; BKV-HC: BK virus-associated hemorrhagic cystitis; CAR: chimeric antigen receptor; CDV: cidofovir; CHASER: cyclophosphamide, cytarabine, etoposide, dexamethasone, and rituximab; CMR: complete metabolic response; CMV: cytomegalovirus; CR: complete response; CT: computed tomography; DLBCL: relapsed/refractory diffuse large B-cell lymphoma; EBV: Epstein-Barr virus; LDH: lactate dehydrogenase; mPSL: methylprednisolone; PBSCH: peripheral blood stem cell harvest; PET/CT: positron emission tomography/computed tomography; PD: progressive disease; PMD: progressive metabolic disease; PSL: prednisolone; R-CHOP: rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone; R-GDP: rituximab plus gemcitabine, dexamethasone, and cisplatin; RR: partial response; Pola-BR: polatuzumab-rituximab-bendamustine combination therapy; sIL-2R: serum soluble interleukin-2 receptor; SOS: sinusoidal obstruction syndrome; T-bil: total bilirubin
| References | ▴Top |
- Tilly H, Morschhauser F, Sehn LH, Friedberg JW, Trneny M, Sharman JP, Herbaux C, et al. Polatuzumab vedotin in previously untreated diffuse large B-cell lymphoma. N Engl J Med. 2022;386(4):351-363.
doi pubmed - Crump M, Neelapu SS, Farooq U, Van Den Neste E, Kuruvilla J, Westin J, Link BK, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study. Blood. 2017;130(16):1800-1808.
doi pubmed - Sehn LH, Herrera AF, Flowers CR, Kamdar MK, McMillan A, Hertzberg M, Assouline S, et al. Polatuzumab vedotin in relapsed or refractory diffuse large B-cell lymphoma. J Clin Oncol. 2020;38(2):155-165.
doi pubmed - Zelenetz AD, Gordon LI, Chang JE, Christian B, Abramson JS, Advani RH, Bartlett NL, et al. NCCN guidelines(R) insights: B-cell lymphomas, Version 5.2021. J Natl Compr Canc Netw. 2021;19(11):1218-1230.
doi pubmed - Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, et al. Axicabtagene ciloleucel car T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-2544.
doi pubmed - Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, Jager U, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380(1):45-56.
doi pubmed - Abramson JS, Palomba ML, Gordon LI, Lunning MA, Wang M, Arnason J, Mehta A, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396(10254):839-852.
doi pubmed - Thieblemont C, Phillips T, Ghesquieres H, Cheah CY, Clausen MR, Cunningham D, Do YR, et al. Epcoritamab, a novel, subcutaneous CD3xCD20 bispecific T-cell-engaging antibody, in relapsed or refractory large B-cell lymphoma: dose expansion in a phase I/II trial. J Clin Oncol. 2023;41(12):2238-2247.
doi pubmed - Dhedin N, Giraudier S, Gaulard P, Esperou H, Ifrah N, Michallet M, Milpied N, et al. Allogeneic bone marrow transplantation in aggressive non-Hodgkin's lymphoma (excluding Burkitt and lymphoblastic lymphoma): a series of 73 patients from the SFGM database. Societ Francaise de Greffe de Moelle. Br J Haematol. 1999;107(1):154-161.
doi pubmed - Lazarus HM, Zhang MJ, Carreras J, Hayes-Lattin BM, Ataergin AS, Bitran JD, Bolwell BJ, et al. A comparison of HLA-identical sibling allogeneic versus autologous transplantation for diffuse large B cell lymphoma: a report from the CIBMTR. Biol Blood Marrow Transplant. 2010;16(1):35-45.
doi pubmed - Cesaro S, Dalianis T, Hanssen Rinaldo C, Koskenvuo M, Pegoraro A, Einsele H, Cordonnier C, et al. ECIL guidelines for the prevention, diagnosis and treatment of BK polyomavirus-associated haemorrhagic cystitis in haematopoietic stem cell transplant recipients. J Antimicrob Chemother. 2018;73(1):12-21.
doi pubmed - Abudayyeh A, Hamdi A, Lin H, Abdelrahim M, Rondon G, Andersson BS, Afrough A, et al. Symptomatic BK Virus Infection Is Associated With Kidney Function Decline and Poor Overall Survival in Allogeneic Hematopoietic Stem Cell Recipients. Am J Transplant. 2016;16(5):1492-1502.
doi pubmed - Kerbauy LN, Kerbauy MN, Bautzer V, Chapchap EC, de Mattos VRP, da Rocha JDA, Esteves I, et al. Severe hemorrhagic cystitis caused by the BK polyomavirus is associated with decreased survival post-allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis. 2019;21(5):e13101.
doi pubmed - Mohammadi Najafabadi M, Soleimani M, Ahmadvand M, Soufi Zomorrod M, Mousavi SA. Treatment protocols for BK virus associated hemorrhagic cystitis after hematopoietic stem cell transplantation. Am J Blood Res. 2020;10(5):217-230.
pubmed - Balfour JA, Goa KL. Bendamustine. Drugs. 2001;61(5):631-638; discussion 639-640.
doi pubmed - Saito H, Maruyama D, Maeshima AM, Makita S, Kitahara H, Miyamoto K, Fukuhara S, et al. Prolonged lymphocytopenia after bendamustine therapy in patients with relapsed or refractory indolent B-cell and mantle cell lymphoma. Blood Cancer J. 2015;5(10):e362.
doi pubmed - Fung M, Jacobsen E, Freedman A, Prestes D, Farmakiotis D, Gu X, Nguyen PL, et al. Increased Risk of Infectious Complications in Older Patients With Indolent Non-Hodgkin Lymphoma Exposed to Bendamustine. Clin Infect Dis. 2019;68(2):247-255.
doi pubmed - Mori Y, Miyamoto T, Kato K, Kamezaki K, Kuriyama T, Oku S, Takenaka K, et al. Different risk factors related to adenovirus- or BK virus-associated hemorrhagic cystitis following allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2012;18(3):458-465.
doi pubmed - Giraud G, Bogdanovic G, Priftakis P, Remberger M, Svahn BM, Barkholt L, Ringden O, et al. The incidence of hemorrhagic cystitis and BK-viruria in allogeneic hematopoietic stem cell recipients according to intensity of the conditioning regimen. Haematologica. 2006;91(3):401-404.
pubmed - Giraud G, Priftakis P, Bogdanovic G, Remberger M, Dubrulle M, Hau A, Gutmark R, et al. BK-viruria and haemorrhagic cystitis are more frequent in allogeneic haematopoietic stem cell transplant patients receiving full conditioning and unrelated-HLA-mismatched grafts. Bone Marrow Transplant. 2008;41(8):737-742.
doi pubmed - Silva Lde P, Patah PA, Saliba RM, Szewczyk NA, Gilman L, Neumann J, Han XY, et al. Hemorrhagic cystitis after allogeneic hematopoietic stem cell transplants is the complex result of BK virus infection, preparative regimen intensity and donor type. Haematologica. 2010;95(7):1183-1190.
doi pubmed - Uhm J, Hamad N, Michelis FV, Shanavas M, Kuruvilla J, Gupta V, Lipton JH, et al. The risk of polyomavirus BK-associated hemorrhagic cystitis after allogeneic hematopoietic SCT is associated with myeloablative conditioning, CMV viremia and severe acute GVHD. Bone Marrow Transplant. 2014;49(12):1528-1534.
doi pubmed - Saade A, Gras J, Darmon M, Michonneau D, Dhedin N, Feghoul L, Le Goff J, et al. Incidence, risk factors and outcome of BK virus hemorrhagic cystitis following allogenic hematopoietic cell transplantation: a retrospective cohort study. Bone Marrow Transplant. 2022;57(8):1287-1294.
doi pubmed - Zhou X, Zhang S, Fan J, Zhu X, Hu S. Risk factors for BK virus-associated hemorrhagic cystitis after allogeneic hematopoietic stem cell transplantation: A systematic review and meta-analysis. Clin Transplant. 2023;37(11):e15121.
doi pubmed - Gilis L, Morisset S, Billaud G, Ducastelle-Lepretre S, Labussiere-Wallet H, Nicolini FE, Barraco F, et al. High burden of BK virus-associated hemorrhagic cystitis in patients undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2014;49(5):664-670.
doi pubmed - Han TT, Xu LP, Liu DH, Liu KY, Fu HX, Zhao XY, Zhao XS, et al. Cytomegalovirus is a potential risk factor for late-onset hemorrhagic cystitis following allogeneic hematopoietic stem cell transplantation. Am J Hematol. 2014;89(1):55-61.
doi pubmed - Bielorai B, Shulman LM, Rechavi G, Toren A. CMV reactivation induced BK virus-associated late onset hemorrhagic cystitis after peripheral blood stem cell transplantation. Bone Marrow Transplant. 2001;28(6):613-614.
doi pubmed - Espada E, Cheng MP, Kim HT, Woolley AE, Avigan JI, Forcade E, Soares MVD, et al. BK virus-specific T-cell immune reconstitution after allogeneic hematopoietic cell transplantation. Blood Adv. 2020;4(9):1881-1893.
doi pubmed - Coomes EA, Wolfe Jacques A, Michelis FV, Kim DDH, Thyagu S, Viswabandya A, Lipton JH, et al. Efficacy of cidofovir in treatment of BK virus-induced hemorrhagic cystitis in allogeneic hematopoietic cell transplant recipients. Biol Blood Marrow Transplant. 2018;24(9):1901-1905.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.