| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Original Article
Volume 13, Number 6, December 2024, pages 261-267
Optimizing Falling Drop Hemoglobin Method by Comparing Capillary Versus Venous Blood and Determining the Stability of the Copper Sulfate Solution
Amogh Chariyamanea, Tim R. Randolpha, b
aDepartment of Clinical Health Sciences, Doisy College of Health Sciences, Saint Louis
University, St. Louis, MO 63104, USA
bCorresponding Author: Tim R. Randolph,
Department of Clinical Health Sciences, Doisy College of Health Sciences, Saint Louis
University, St. Louis, MO 63104, USA
Manuscript submitted August 29, 2024, accepted October 31, 2024, published online December 2,
2024
Short title: Optimizing Falling Drop Hemoglobin Method
doi:
https://doi.org/10.14740/jh1337
| Abstract | ▴Top |
Background: Anemia is a global health issue that affects over 1 billion people and contributes to maternal mortality and birth defects. Low-resource, tropical areas face a dual challenge: high prevalence of anemia and inability to access affordable testing methods. The falling drop hemoglobin method has been developed by our lab to quantify hemoglobin concentration and assess anemia by timing the descent of venous blood in a column of copper sulfate solution, without using electricity or batteries. This research aimed to optimize the falling drop hemoglobin method by evaluating the use of capillary blood to reduce within sample variance and assessing copper sulfate stability to determine shelf life in expected working conditions.
Methods: The falling drop hemoglobin method was performed on both venous and capillary blood samples collected directly from the fingertip by dispensing 44 µL of blood in a copper sulfate column. A microhematocrit was performed on the venous blood sample and converted mathematically to a hemoglobin level to serve as the standard. Copper sulfate stability was assessed for 32 weeks among three solutions: solution prepared fresh on day of testing, solution incubated at room temperature, and solution incubated at 37.7 °C.
Results: Capillary blood yielded higher average descent times and higher standard deviations than venous blood. Collecting precisely 44 µL of capillary blood proved challenging and impractical. In copper sulfate stability testing, freshly prepared solution yielded the highest average descent time. A one-way analysis of variance (ANOVA) test and Tukey’s honestly significant difference (HSD) post hoc testing revealed no significant difference between mean descent times of freshly prepared and 37.7 °C solutions (P = 0.26) and between room temperature and 37.7 °C solutions (P = 0.64), but a significant difference between freshly prepared and room temperature solutions (P = 0.04).
Conclusions: This study found that capillary blood did not present a more accurate alternative to venous blood in the falling drop hemoglobin test, and copper sulfate did not degrade over 32 weeks at 37.7 °C. This lends support for the current use of venous blood in the test, and for use of copper sulfate solution in tropical climates, where the test is most necessary, with a shelf life of at least 32 weeks.
Keywords: Hemoglobin; Hematocrit; Test; Anemia; Underdeveloped; Resource-poor; Developing
| Introduction | ▴Top |
Anemia is a significant global health problem that affects billions of individuals and is most notably persistent in developing nations, particularly in tropical regions. Anemia is a reduction in the oxygen-carrying capacity of the blood and is broadly characterized by an insufficiency of red blood cells (RBCs) and the oxygen-carrying molecule, hemoglobin, contained within. Hundreds of causes of anemia exist that can be divided into three categories based on RBC size: microcytic, normocytic, and macrocytic. Iron deficiency is a common cause of microcytic anemia and is believed to account for at least 75% of anemia cases. Besides iron deficiency, infection and hemoglobinopathies (both normocytic) are common causes of this health condition in developing countries [1]. Common causes of macrocytic anemia include deficiencies of vitamin B12 and folate. Both mild and severe forms of anemia are prevalent [2]. Severe, often life-threatening forms of anemia include malarial anemia, sickle cell disease, aplastic anemia (all normocytic), and severe thalassemia (microcytic), all of which are prevalent in the developing world [2]. Although mild forms of anemia are more common, severe cases can lead to increased risk of maternal and child mortality, cognitive and physical underdevelopment in children, and limited physical capabilities of adults [3].
The adverse effects of anemia support the need to effectively diagnose and monitor this condition. The complete blood count (CBC) contains three parameters used to identify anemia: RBC count, hemoglobin, and hematocrit, with hemoglobin and hematocrit being the most commonly used. There currently exist several methods to determine hemoglobin levels, including the gravimetric copper sulfate, cyanmethemoglobin, azide-methemoglobin, optical reflectance technology, and color scale methods.
A common method used to screen potential blood donors for hemoglobin concentrations is the copper sulfate method. A drop of capillary blood from the fingertip is allowed to fall into a container of copper sulfate solution with known specific gravity. The blood hemoglobin concentration is deemed sufficient for blood donation only if the droplet sinks to the bottom of the container within 15 s, showing that blood hemoglobin concentration meets the 12.5 g/dL cut-off for female donors in the United States and the Council of Europe. Blood hemoglobin concentration requirements for male donors can be up to 1 g/dL higher than those for female donors. Although relatively easy to perform and inexpensive, this test does not quantify hemoglobin concentration, which is needed for accurate anemia diagnosis [4].
One of the most accurate quantitative hemoglobin methods is the cyanmethemoglobin method that employs spectrophotometry and has historically been used in hematology analyzers. This measurement is performed by first hemolyzing RBCs and then adding potassium ferricyanide to produce methemoglobin. Cyanmethemoglobin is then created by mixing the methemoglobin with potassium cyanide. The absorbance of this stable product is measured at 540 nm and compared against a standard solution, allowing for hemoglobin level determination [5].
There are several very similar alternatives to the cyanmethemoglobin method that are less toxic and thus preferred for use in automated hematology analyzers. One such method uses alkaline hematin or alkaline borate instead of a cyanide reagent. In the alkaline hematin method, a basic solution that contains a non-ionic detergent compound (Triton X-100) is added to a small sample of blood. The subsequent reactions hemolyze the RBCs and produce a stable compound from the hemoglobin derivatives in the sample. Absorbance is measured at 575 nm. These alternative methods use compounds that are less toxic and often cheaper than cyanide reagents and yet can still work to accurately measure hemoglobin concentrations in automated instruments [6].
Another popular version is the azide-methemoglobin method employed by the HemoCue point-of-care instrument. In this method, the RBCs are hemolyzed and reacted with sodium nitrite to convert hemoglobin to methemoglobin, which is combined with sodium azide to yield azide-methemoglobin. A spectrophotometer is used to measure absorbance at two wavelengths of 565 nm and 880 nm to counteract the effects of turbidity in the sample, which would falsely elevate the hemoglobin measurement. The HemoCue system is a small, portable device that only measures hemoglobin and can be operated using electricity or batteries [7].
Another less expensive option for hemoglobin measurement, which also does not require large, complex instruments, utilizes optical reflectance technology. In the optical reflectance system, blood samples are placed on test strips that contain erythrocyte lysing agents. In the Seralyzer reflectance photometer, saponin is used to lyse RBCs, and potassium ferricyanide is used to convert hemoglobin to more stable methemoglobin [8]. In the Mission Plus Hemoglobin meter, RBC lysis is followed by conversion of hemoglobin to azide-methemoglobin [9, 10]. Optical reflectance technologies then use a battery operated, handheld reflectance meter to analyze the color intensity of reflected light from the test strip and thereby quantify the total hemoglobin concentration in the sample. This measurement is compared to a baseline optical reflectance to calculate total hemoglobin concentration.
Finally, the World Health Organization’s visual method uses a six-point, fixed color scale to compare the color of the patient’s blood placed on absorbent test papers. Darker colors indicate higher hemoglobin levels. Like the reflectance method, the color scale method does not require electricity, but its hemoglobin measurements are much less precise. This is due to the reliance on the subjectivity of visual assessment of color, as well as the color scale representing six fixed color spots, defined as 4, 6, 8, 10, 12, and 14 g/dL. Therefore, results could potentially be different by as much as 2 - 3 g/dL from the actual concentration [11].
In developing countries, clinics often lack finances, stable electricity, refrigeration, climate control, clean running water, and other important resources needed to support hematology instruments that measure hemoglobin [12]. Due to these limitations, the modern hemoglobin testing methods are difficult and impractical to implement in developing countries where anemia diagnosis is most critical. Cell counting instruments that perform CBCs, while common in modern labs, are uncommon in the developing world because they are cost prohibitive and require stable electricity, as well as specialized training to operate, maintain, and repair. Most also use cyanide or azide-based reactions to measure hemoglobin that require careful waste disposal. Point-of-care instruments like the HemoCue system and reflectance devices are a fraction of the cost of a cell counting instrument but the consumables (cuvettes and test strips) are often cost prohibitive to many rural labs in developing countries. The HemoCue system uses azide that requires proper disposal, and all point-of-care methods discussed require electricity or batteries. Although simpler tests, such as the copper sulfate method or visual color method, do not require electricity or expensive instruments, neither measures hemoglobin concentration to the level of precision required to diagnose and care for anemic patients. The copper sulfate method does not provide a quantitative measurement of hemoglobin level, and the visual color method at best provides an estimate of the hemoglobin level. These shortcomings highlight the need for an inexpensive yet accurate hemoglobin measurement method that is accessible to the developing world.
A simple, inexpensive, and electricity-free test has been developed in our lab, the falling drop hemoglobin method, which is a modification of the copper sulfate method used to screen potential blood donors in blood collection facilities. This method quantifies hemoglobin levels based on the descent time of a drop of blood through a copper sulfate column [13]. The falling drop hemoglobin method demonstrates a linear relationship between the average descent time of six successive venous blood drops and hemoglobin level. However, reducing the number of drops required for each test would optimize the method, and there is a need to test the stability of the copper sulfate solution under conditions that simulate those in clinics in developing countries.
The purpose of this study is to test the falling drop hemoglobin method using capillary blood, with a goal of improving the test’s coefficient of determination (r2) and reducing the number of trials per test. Additionally, tests will be performed using copper sulfate at varying temperatures and duration times to determine the stability of the copper sulfate solution under conditions that mimic open-air labs in developing countries.
| Materials and Methods | ▴Top |
Falling drop hemoglobin method
The falling drop hemoglobin method was performed as described in previous publications as follows:
Column assembly
The central apparatus for the falling drop hemoglobin method consists of a modified erythrocyte sedimentation rack, which holds five glass columns. Each glass column is 61 cm long, 10 mm outer diameter (OD) and 7.7 mm inner diameter (ID). The bottom end was sealed with silicone with two marked lines at the top spaced 3 cm apart (the top mark is the copper sulfite fill line and the lower is the start timer mark), and one line about 2 cm from the bottom, the stop timer mark [13]. Five LABSCO, 2 and 13/16, 200 - 1,000 µL pipet tips were modified via removal of 3 cm from the bottom tip and placed on top of the columns to stabilize the pipet during delivery of blood droplets [14].
Reagent preparation
Copper sulfate pentahydrate (Acros Organics, New Jersey) was dissolved in deionized water to produce a 4.58% copper sulfate solution. A refractometer was used to verify that the solution’s specific gravity measured 1.015. Each column was adjusted to stand upright in the modified rack and then filled to the upper mark with the 4.58% copper sulfate solution [14]. Figure 1 depicts the set-up for the falling drop hemoglobin method.
 Click for large image |
Figure 1. Components of the falling drop hemoglobin method, showing only one glass column. |
Sample collection
One sodium fluoride/potassium oxalate anticoagulant tube of venous blood was collected using standard venipuncture technique. Previous experiments showed improved blood drop stability and reduced variance when using oxalate compared to ethylenediaminetetraacetic acid (EDTA), sodium citrate, and sodium heparin [14]. Capillary blood was drawn using standard capillary puncture technique, and droplets were collected directly into a variable volume AlphaPette automated pipettor that was set to 44 µL using the modified tip described above. All blood samples were collected from members of the research team, negating the need for the Institutional Review Board (IRB) approval. The venous and capillary samples were collected from the same research team member. This study was conducted in compliance with the ethical standards of the responsible institution on human subjects.
Testing of capillary and venous blood
Forty-four microliters of either venous or capillary blood were dispensed onto the copper sulfate solution surface within the glass column. The descent time of the blood droplet was measured in seconds. Qualitative observations of the blood droplet, including its morphology (plasma tails and wobble) and trajectory, were also recorded. To reuse a column for a second test, 1 mL of copper sulfate solution was removed from the top of the glass column to remove residual blood and replaced with fresh solution. After testing, a microhematocrit was performed on the venous blood sample and compared to a standard curve created in our lab for the venous method to ensure ongoing stability and reproducibility.
Copper sulfate stability testing
Two separate, 1-L containers of 4.58% copper sulfate solution were prepared. One was incubated at room temperature, and one was incubated at 37.7 °C. A third batch of copper sulfate solution was prepared fresh on each day of testing. During each trial, spaced 1 week apart, the falling drop hemoglobin method was performed using venous blood on each of the three copper sulfate solutions. The descent time of the six blood droplets per test was measured in seconds and averaged to generate the test result. Average descent times and qualitative observations of the blood droplets were also recorded.
Data analysis
Average descent time and standard deviation (SD) were calculated for each capillary and venous blood sample. These values, along with qualitative observations of droplet morphology (plasma tails and wobble) and trajectory for the blood droplets, were compared across the two blood samples. The corresponding hematocrit levels, based on a descent time compared to a standard curve, were compared to the true measured microhematocrit of the blood sample, to determine accuracy.
Average descent times and SD for the venous blood samples from the copper sulfate stability testing were calculated. Average descent times were plotted against time in weeks with 95% confidence intervals to compare the three different copper sulfate solutions over time. Mean descent times were compared using a one-way analysis of variance (ANOVA) test and a Tukey’s honestly significant difference (HSD) test.
| Results | ▴Top |
Comparing capillary and venous blood
Blood droplet descent times in the falling drop hemoglobin method were compared between capillary and venous blood. Table 1 shows average and SD of the descent time, as well as corresponding average hematocrit, for both capillary and venous blood. Capillary blood yielded higher average descent time and higher SD than venous blood. This resulted in the falling drop hemoglobin method measuring lower hematocrit values when capillary blood was used.
 Click to view |
Table 1. Comparison of Capillary and Venous
Blood in the Falling Drop Hemoglobin Method |
Collecting and testing capillary blood in the falling drop hemoglobin method proved challenging compared to venous blood. The collection of capillary blood droplets directly into the pipette tip often led to smearing of the blood droplet at the puncture site and inclusion of air bubbles. This resulted in unsuccessful collection or unequal droplet sizes (less than 44 µL) between trials. Capillary puncture occasionally required repeating on a second finger to obtain the six droplets of blood needed for a single result. Time constraints, due to clotting at the puncture site, often led to rushed delivery of the capillary blood droplet, resulting in oddly shaped or fragmented droplets within the glass column.
Testing copper sulfate stability
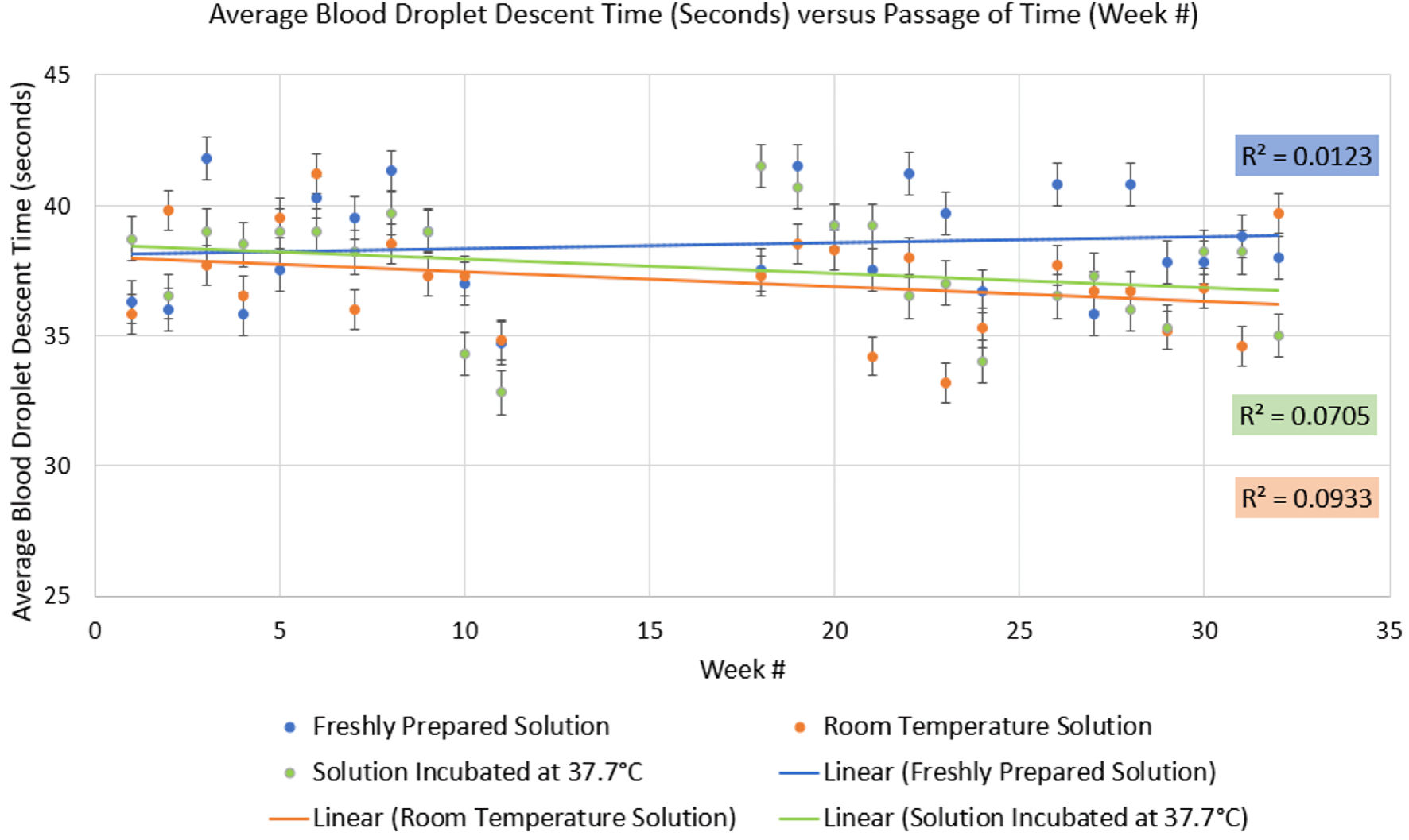
Blood droplet descent times were collected 1 week apart for 32 weeks using three solutions of copper sulfate: freshly prepared solution, room temperature incubated solution, and 37.7 °C incubated solution. Table 2 depicts average, range, and SD of descent times among each solution. Room temperature solution yielded the lowest SD, while freshly prepared solution and solution incubated at 37.7 °C yielded similar, yet slightly higher SDs. Figure 2 depicts changes in droplet descent time over a 32-week period using three copper sulfate solutions. Descent times for all three solutions changed over time to similar extents, with no clear shift. However, from Trial 8 onwards, the 37.7 °C incubated solution exhibited a slight downward trend in descent time. A one-way ANOVA showed no statistically significant difference between the mean descent times of the three solutions, with a P value greater than 0.05 (P = 0.050081). Since the calculated P value was only marginally above the significance threshold, Tukey’s HSD post hoc test was completed. The results showed a statistically significant difference between the mean descent times of freshly prepared and room temperature solutions (P = 0.04), no statistically significant difference between freshly prepared and 37.7 °C solutions (P = 0.26), and no statistically significant difference between room temperature and 37.7 °C solutions (P = 0.64).
 Click to view |
Table 2. Copper Sulfate Stability
Testing |
 Click for large image |
Figure 2. Linear regression of blood droplet descent time compared over trials spaced 1 week apart. |
| Discussion | ▴Top |
The goals of this study were twofold: 1) to determine if the use of capillary blood, as is used in blood collection centers employing the copper sulfate screening method, would lower the SD between trials to reduce the number of trials needed per test; and 2) to evaluate the stability of copper sulfate in a variety of conditions to establish its shelf life. Capillary blood yielded higher average descent times and higher SDs than venous blood. This corresponded to a 5.75% lower average hematocrit result using capillary blood, which is clinically significant. Furthermore, collecting precisely 44 µL of capillary blood directly from the fingertip proved challenging. Smearing of blood on the fingertip and inclusion of air bubbles in the pipetted sample reduced the blood volume to less than 44 µL. Less blood introduced into the test system explains the slower descent or higher times and the correspondingly lower hemoglobin (hematocrit) values compared to venous blood. Therefore, capillary blood does not offer a more accurate alternative for the falling drop hemoglobin method.
The copper sulfate stability testing showed that freshly prepared solution had the highest average descent time, while room temperature incubated solution and the solution incubated at 37.7 °C were similar but showed slightly lower average descent times. SD ranged from 3.06 to 4.20 s, with room temperature having the lowest SD and the 37.7 °C solution having the highest. The linear regression curve demonstrated low R2 values suggesting the data are poorly explained by the linear regression model. Nonetheless, the regression lines are relatively flat indicating very little change on average descent time over the 32-week period. There was no clear or drastic point of degradation in any of the three solutions producing a shift in the data. The slightly lower descent time of the room temperature and the 37.7 °C incubated solutions equate to a 1% difference in hematocrit which is not clinically significant. To account for the weakness of the linear regression model, statistical testing was used to compare the mean descent times among the solutions. A one-way ANOVA followed by Tukey’s HSD test revealed that there was a significant difference among the mean descent times of freshly prepared and room temperature solutions, but not between freshly prepared and 37.7 °C solutions or between room temperature and 37.7 °C solutions. While the significant difference between freshly prepared and room temperature solutions was surprising, this is likely due to the variance that is inherent to the falling drop hemoglobin method. Overall, the study found that copper sulfate did not degrade appreciably over 32 weeks at the 37.7 °C designed to mimic ambient temperature in tropical climates.
While the falling drop hemoglobin method offers a hemoglobin testing alternative that is low-cost and electricity-independent, there are limitations to widespread adoption. The primary limitations involve manufacturing of the test kits, training of lab staff, and the redundant requirement of six trials for each hemoglobin assay.
The test kits would contain three key components: the glass columns, the column rack, and the copper sulfate solution. The glass columns used in this prototype were made by cutting long glass tubes to specific lengths and heating them to form a small, sealable tip on one end. This process is feasible to scale to higher levels. The column rack can be crafted from wood and held together with screws. The goal would be to hire a carpenter subcontractor, living in the same locality as the distributor, to create the racks as they are ordered. Part of this mission is to provide gainful employment to citizens of the communities using this method. Copper sulfate can be made in large batches, aliquoted in varying volumes, and included in the test kit to accommodate a variety of usage levels.
Staff training would need to accompany the sale of kits. The vision of this project is to establish distributors of the falling drop hemoglobin method, and other methods that have been developed by the author, to offer sales, training, and advice to customers. Through these avenues, the falling drop hemoglobin method is expected to retain its practicality and be manufactured and utilized where most needed.
The redundant and cumbersome requirement of six drops of blood for each hemoglobin assay reflects the natural variance inherent in the system and an issue that was unable to be overcome. The initial hope was that non-anticoagulated whole capillary blood might improve the inter-run variance, but collecting and delivering a capillary sample to the column proved impractical. It is now hoped that the method’s target beneficiary - clinical labs with limited resources and no electricity - will see the value of a low-cost and electricity-free hemoglobin measurement system and adapt to the need to perform six runs for each hemoglobin test.
In summary, this research supports the use of oxalate anticoagulated venous blood [14] over capillary blood in the falling drop hemoglobin method and the use of copper sulfate solution in tropical climates (37.7 °C) for a 32-week period, where the method is most needed. Future research will focus on the development of a low-cost automated timing system to optimize the efficiency of the falling drop hemoglobin method.
Acknowledgments
None to declare.
Financial Disclosure
The study was funded by the Jack DeLoss Taylor Charitable Trust. Financial support was also received from the DeNardo Education and Research Foundation, and the Department of Clinical Health Sciences at Saint Louis University.
Conflict of Interest
None to declare.
Informed Consent
The study was IRB waived.
Author Contributions
Dr. Tim R. Randolph was the principal investigator of the study, and the one who planned out all facets of the study and was in direct charge of the lab and equipment. Dr. Tim Randolph is also the one who first developed the falling drop hemoglobin method and published this method in 2017. Amogh Chariyamane collected data and performed experiments for the capillary versus venous blood testing and copper sulfate stability testing. Amogh Chariyamane was also the primary writer of the contents of the paper, while Dr. Tim Randolph served as editor and adviser throughout the writing process.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Chaparro CM, Suchdev PS. Anemia epidemiology,
pathophysiology, and etiology in low- and middle-income countries.
Ann N Y Acad Sci. 2019;1450(1):15-31.
doi pubmed - Milman N. Anemia—still a major health problem in many
parts of the world! Ann Hematol. 2011;90(4):369-377.
doi pubmed - Smith C, Teng F, Branch E, Chu S, Joseph KS. Maternal and
perinatal morbidity and mortality associated with anemia in pregnancy. Obstet Gynecol.
2019;134(6):1234-1244.
doi pubmed - Gomez-Simon A, Navarro-Nunez L, Perez-Ceballos E, Lozano ML,
Candela MJ, Cascales A, Martinez C, et al. Evaluation of four rapid methods for hemoglobin
screening of whole blood donors in mobile collection settings. Transfus Apher Sci.
2007;36(3):235-242.
doi pubmed - Srivastava T, Negandhi H, Neogi SB, Sharma J, Saxena R. Methods for hemoglobin estimation: a review of what works. J Hematol Transfus. 2014;2(3):1028-1035.
- Anchinmane VT, Sankhe SV. Evaluation of hemoglobin
estimation with noncyanide alkaline haematin D- 575 method. Int J Res Med Sci.
2016;4:4297-9.
doi - Shiraz A, Debroy A. Haemoglobin screening methods in blood donors - where do we stand now? National Journal of Laboratory Medicine. 2019;8(2):1-4.
- Lott JA, Khabbaza E. Haemoglobin analysis on whole blood by
reflectance photometry. J Automat Chem. 1985;7(4):197-200.
doi pubmed - An R, Huang Y, Man Y, Valentine RW, Kucukal E, Goreke U,
Sekyonda Z, et al. Emerging point-of-care technologies for anemia detection. Lab Chip.
2021;21(10):1843-1865.
doi pubmed - Sahoo J, Epari V, Panigrahi SK, Prasad D, Bhola RK, Mohanty
S, Behera BK. Challenges in detection of adolescent anaemia: validation of point-of-care device
(Mission® plus) for haemoglobin measurement among tribal residential school children of
selected districts of Odisha, India. Indian J Community Med.
2021;46(4):680-684.
doi - Stott GJ, Lewis SM. A simple and reliable method for
estimating haemoglobin. Bull World Health Organ. 1995;73(3):369-373.
pubmed - Veeken H. Hope for Haiti? BMJ.
1993;307(6899):312-313.
doi pubmed - Rupprecht WL, Randolph TR. Improving accuracy of falling drop hemoglobin method via modifications to column length. FASEB J. 2018;32(1 suppl).
- Stumpf MS, Sinha R, Golob N, Hoerner R, Chen C, Randolph TR. Development of a falling drop hemoglobin method for resource-poor settings. Clin Lab Sci. 2017;30(3):131.
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Hematology is published by Elmer Press Inc.