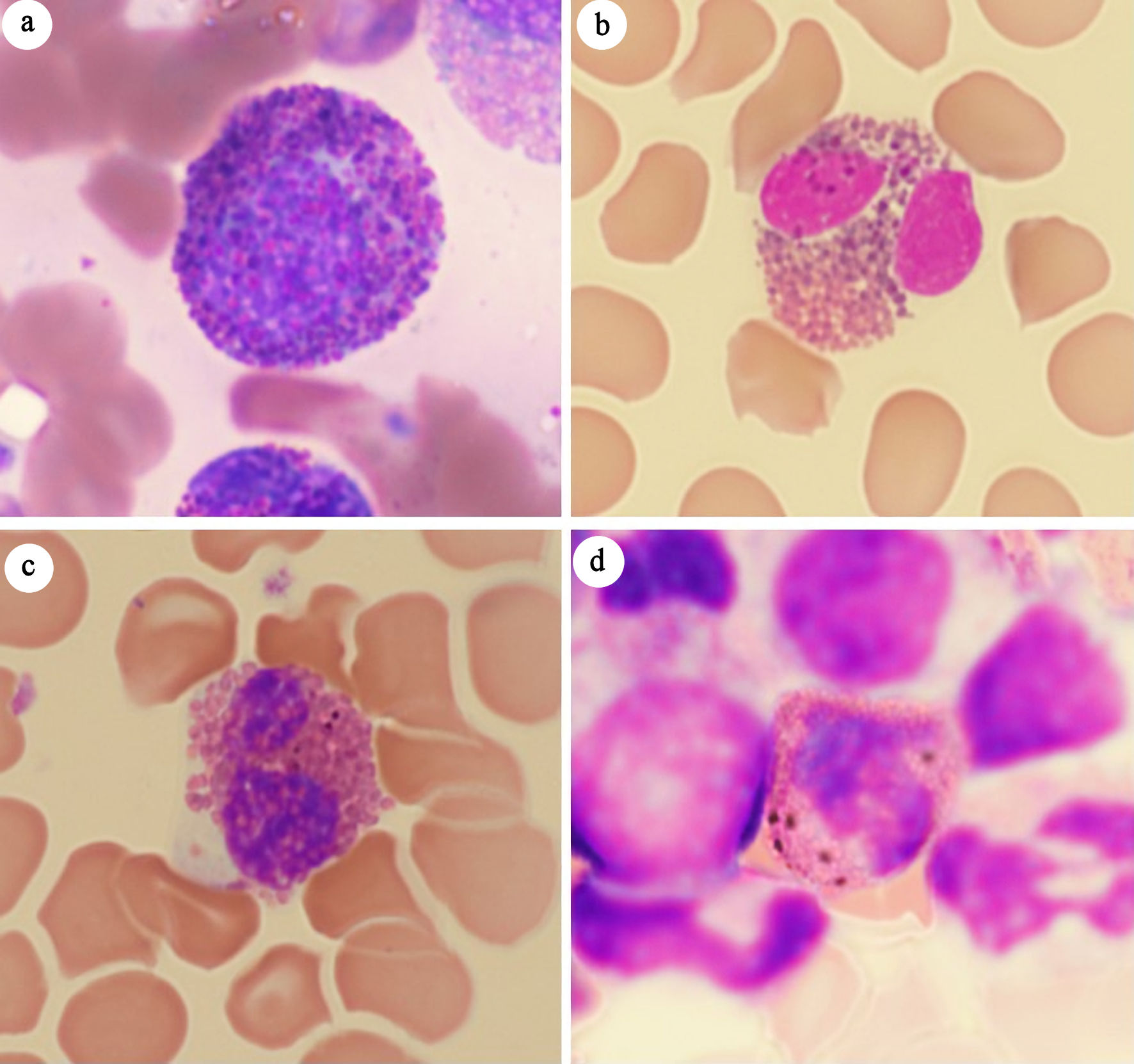
↓ Figure 1. Mimics of Harlequin cells. (a) Eosinophilic myelocyte in reactive bone marrow. (b) Eosinophil with purplish-orange granules. (c) Eosinophil with black granules. (d) Eosinophil with brown granules.
| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Original Article
Volume 15, Number 2, April 2026, pages 99-107
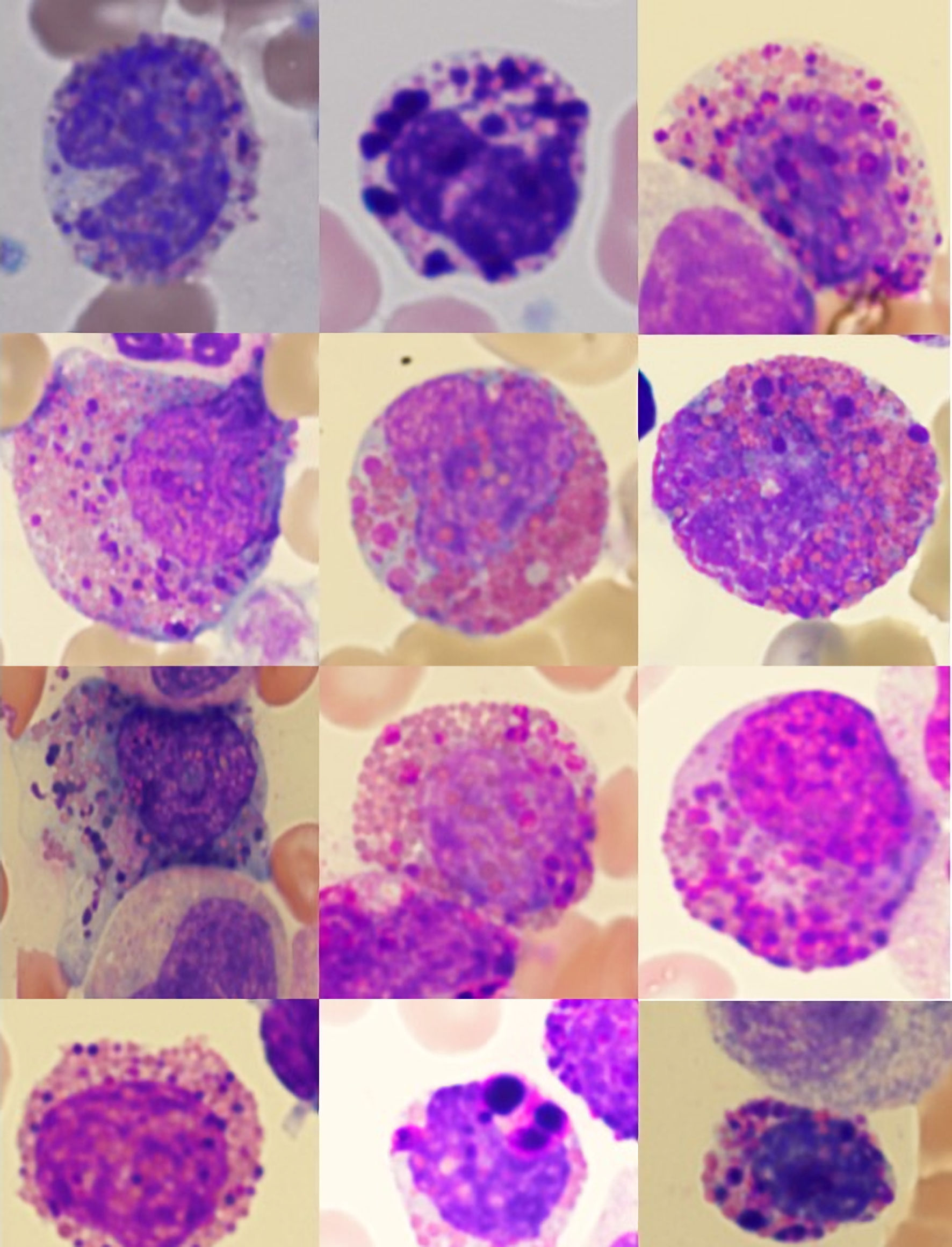
Abnormal Eosinophils With Large, Distinctly Basophilic Granules (Harlequin Cells) on Peripheral Blood Smear: A Clue for Diagnosing Chronic Myeloid Leukemia
Figures

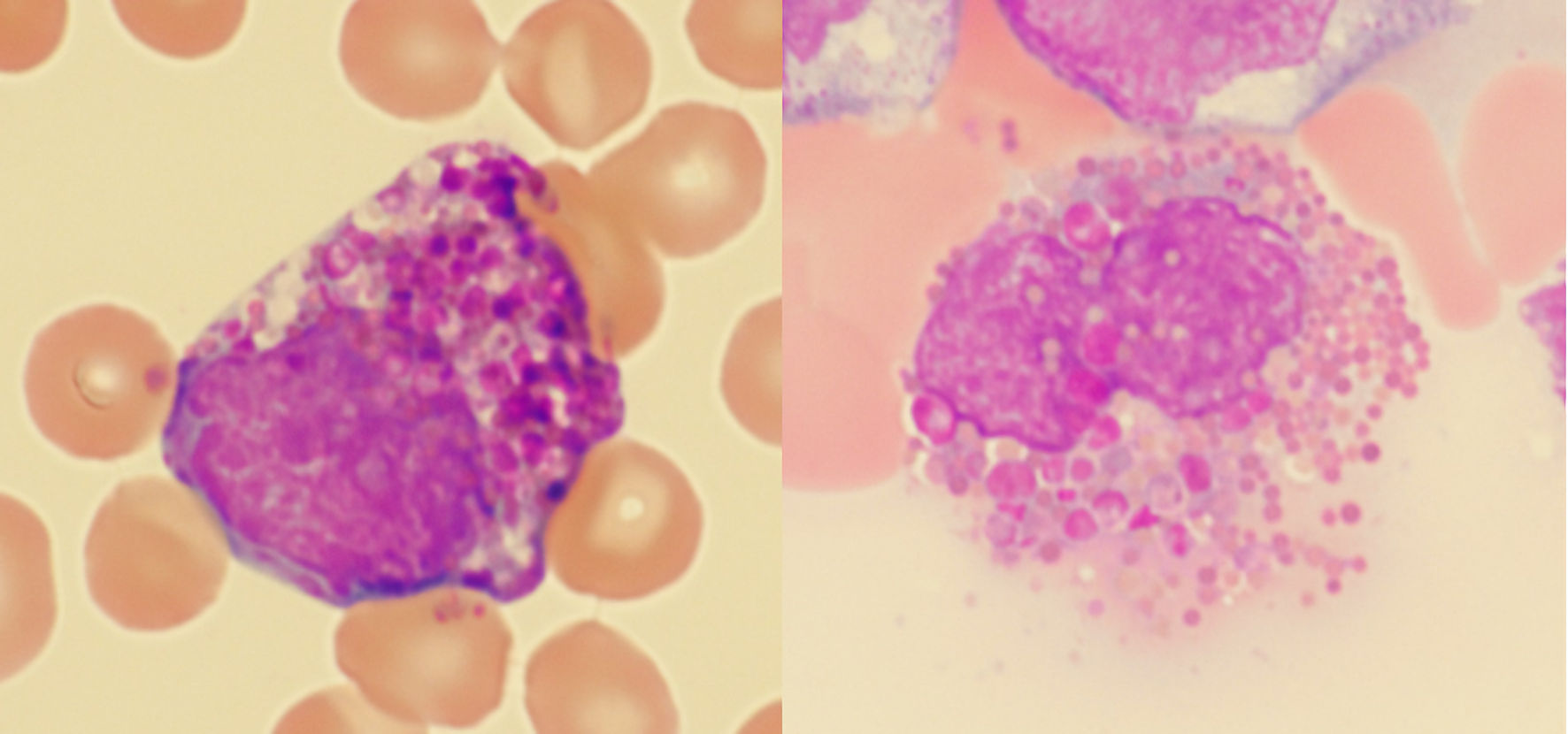
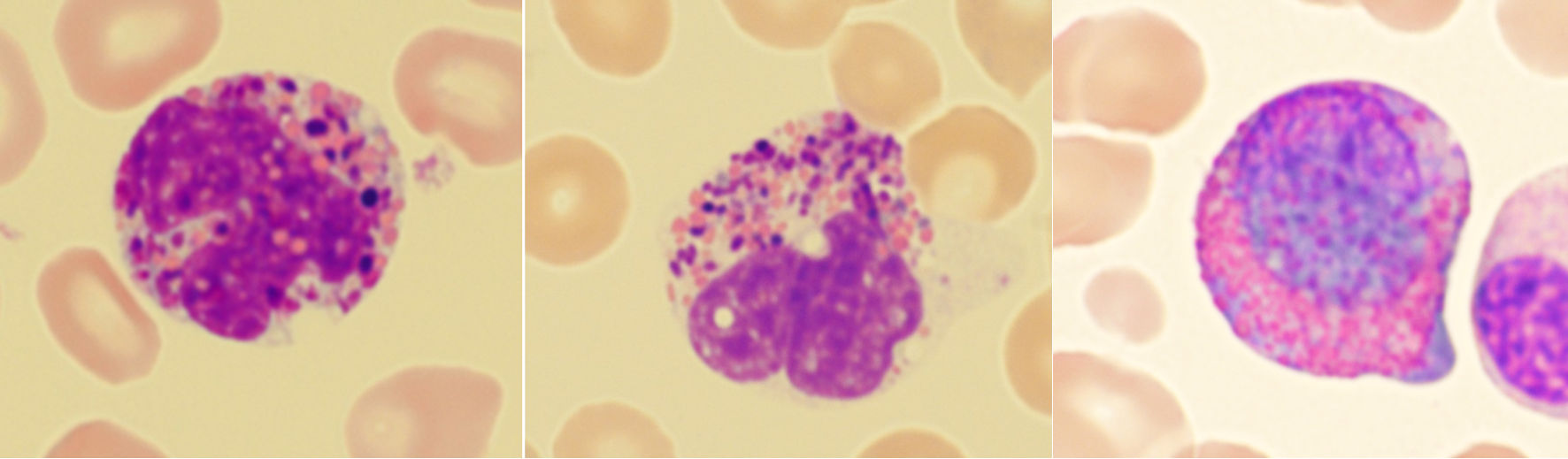
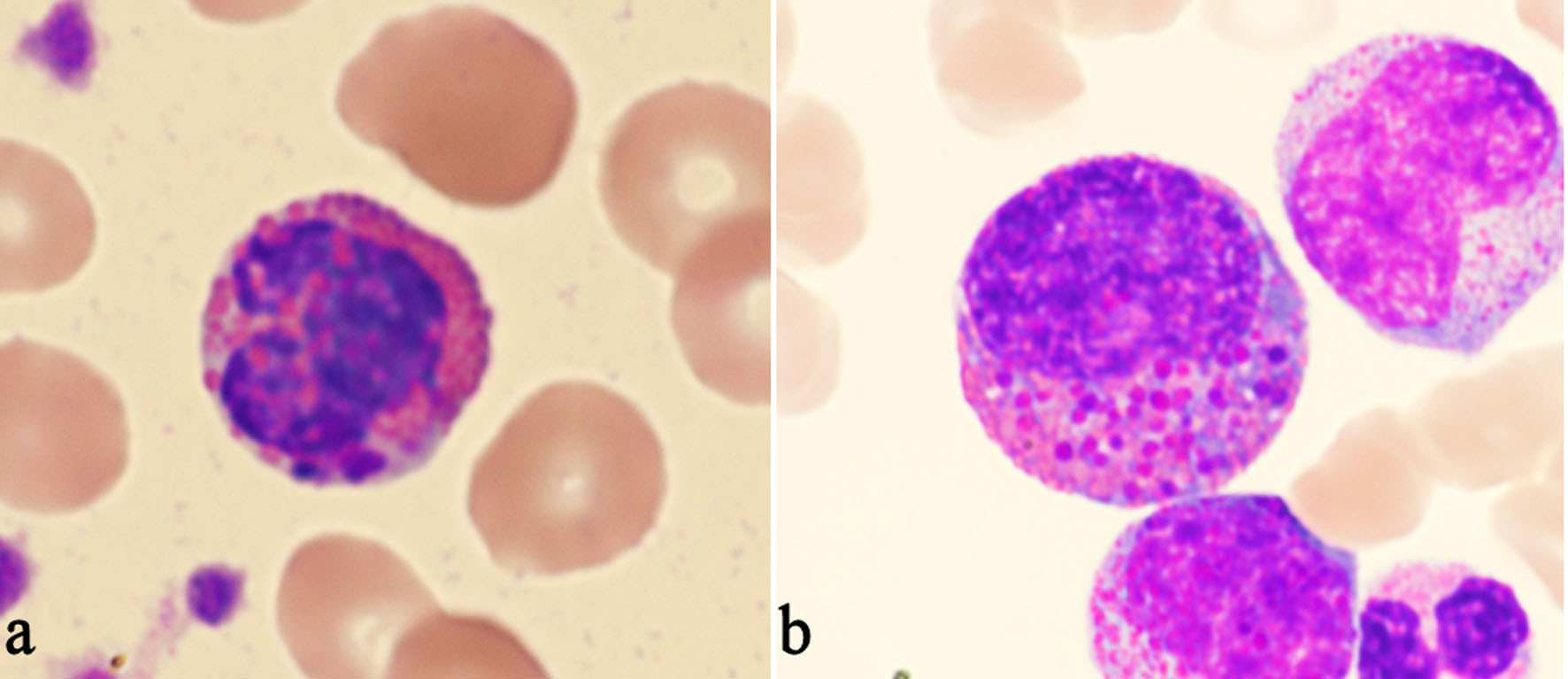
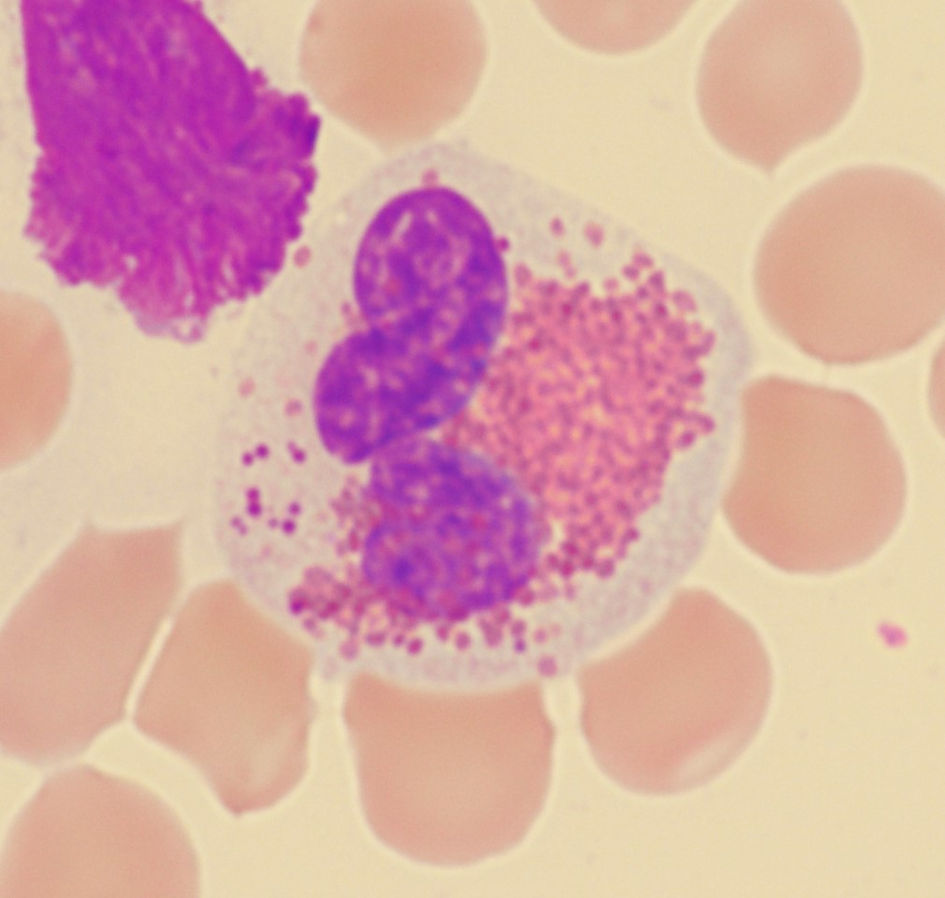
Tables
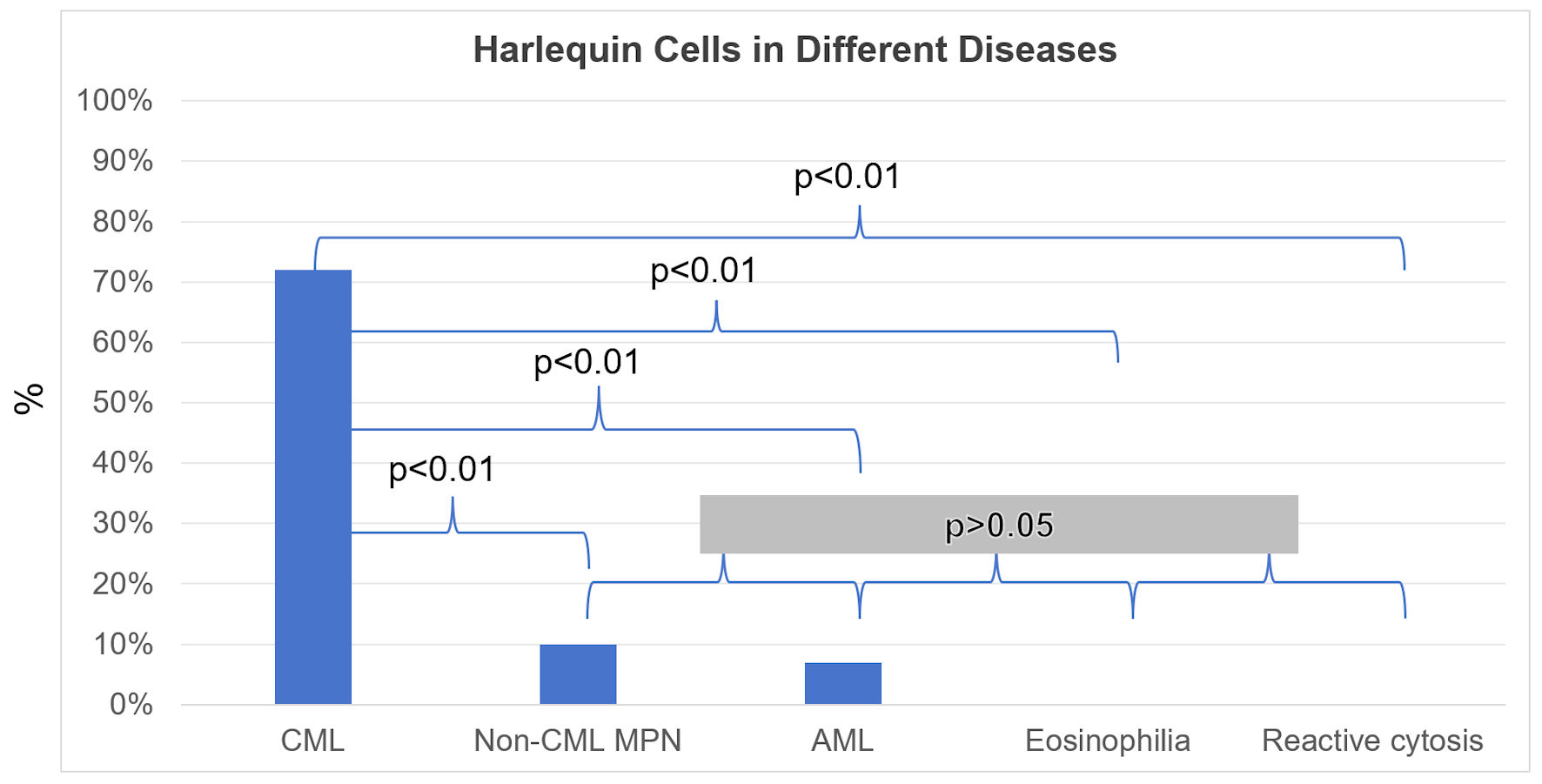
| Group | Diagnosis/phase | Number of cases | Incidence of Harlequin cells, n (%) of cases |
|---|---|---|---|
| Chronic myeloid leukemia (CML) (n = 53) | Chronic phase | 44 | 33 (75%) |
| Accelerated phase | 7 | 4 (57%) | |
| Blast phase | 2 | 1 (50%) | |
| Total | 53 | 38 (72%) | |
| Non-CML myeloproliferative neoplasm and related disorders (n = 30) | Polycythemia vera | 5 | 0 (0%) |
| Essential thrombocythemia | 5 | 0 (0%) | |
| Primary myelofibrosis | 15 | 3 (20%) | |
| SH2B3-associated erythrocytosis | 1 | 0 (0%) | |
| Chronic myelomonocytic leukemia | 4 | 0 (0%) | |
| Total | 30 | 3 (10%) | |
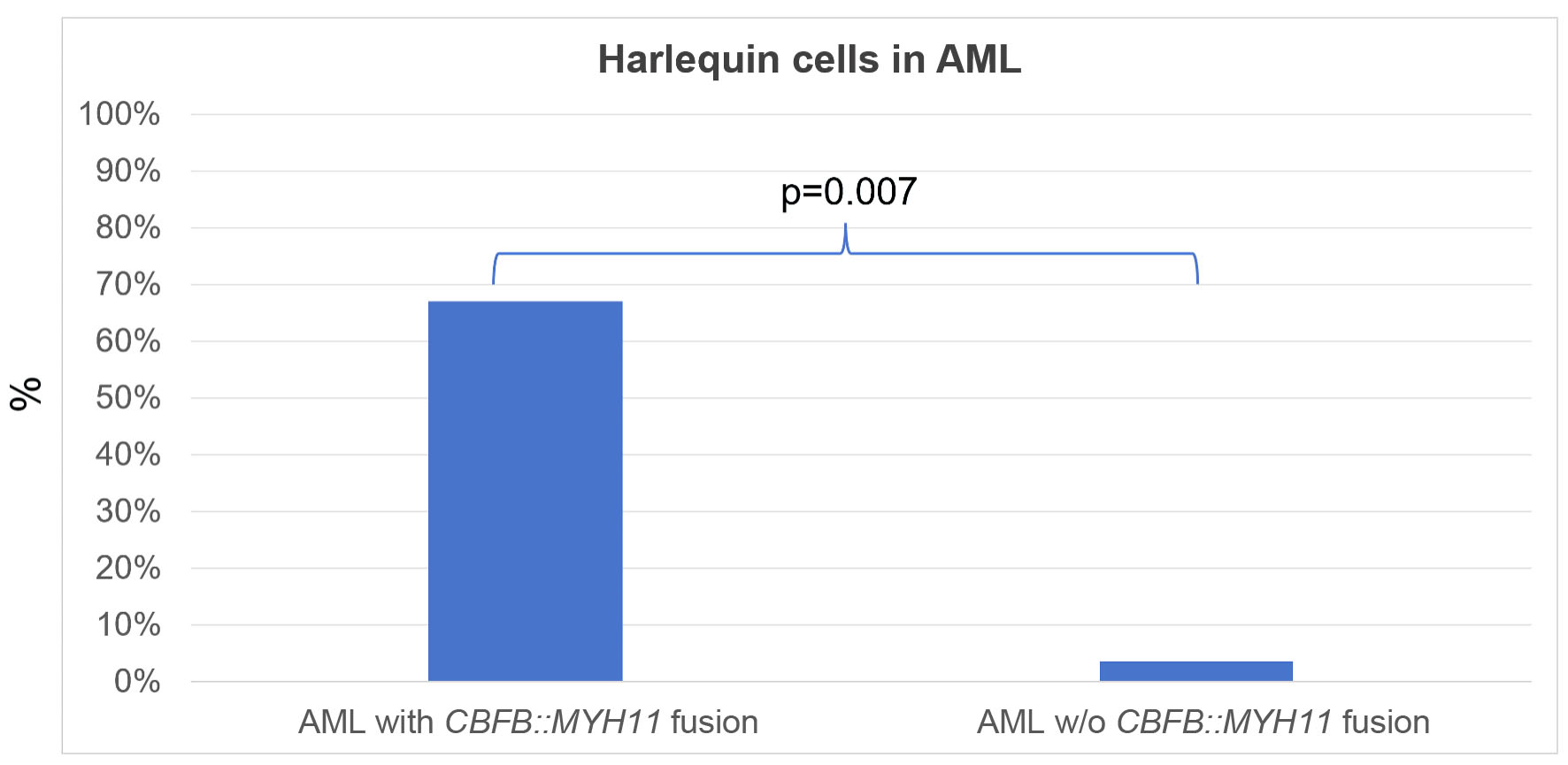
| Acute myeloid leukemia (AML) (n = 59) | AML with CBFB::MYH11 fusion | 3 | 2 (67%) |
| AML with GATA2::MECOM fusion | 2 | 1 (50%) | |
| AML with DEK::DUP214 fusion | 1 | 0 (0%) | |
| AML with KMT2A rearrangement | 2 | 0 (0%) | |
| AML with RUNX1::RUNX1T1 fusion | 4 | 0 (0%) | |
| AML with NPM1 mutation | 10 | 0 (0%) | |
| AML, myelodysplasia-related | 17 | 0 (0%) | |
| AML defined by differentiation | 15 | 1 (7%) | |
| AML post cytotoxic therapy | 5 | 0 (0%) | |
| Total | 59 | 4 (7%) | |
| Eosinophilia (n = 11) | Myeloid neoplasm with PDGFRA rearrangement | 2 | 0 (0%) |
| Lymphocytic variant of hypereosinophilic syndrome | 1 | 0 (0%) | |
| Idiopathic hypereosinophilic syndrome | 6 | 1 (17%) | |
| Reactive eosinophilia | 2 | 0 (0%) | |
| Total | 11 | 1 (9%) | |
| Reactive cytosis (n = 24) | Erythrocytosis/polycythemia | 8 | 0 (0%) |
| Thrombocytosis | 6 | 0 (0%) | |
| Leukemoid reaction | 7 | 0 (0%) | |
| Leukocytosis + thrombocytosis | 3 | 0 (0%) | |
| Total | 24 | 0 (0%) |
| CML | Non-CML myeloproliferative neoplasm and related disorders | AML | Eosinophilia | Reactive cytosis | |
|---|---|---|---|---|---|
| CML: chronic myeloid leukemia; AML: acute myeloid leukemia. | |||||
| Age (years), median (range) | 47 (9–75) | 65 (20–89) | 54 (16–83) | 42 (6–89) | 55 (20–88) |
| Sex, n | |||||
| Male | 33 | 17 | 38 | 6 | 16 |
| Female | 20 | 13 | 21 | 5 | 8 |