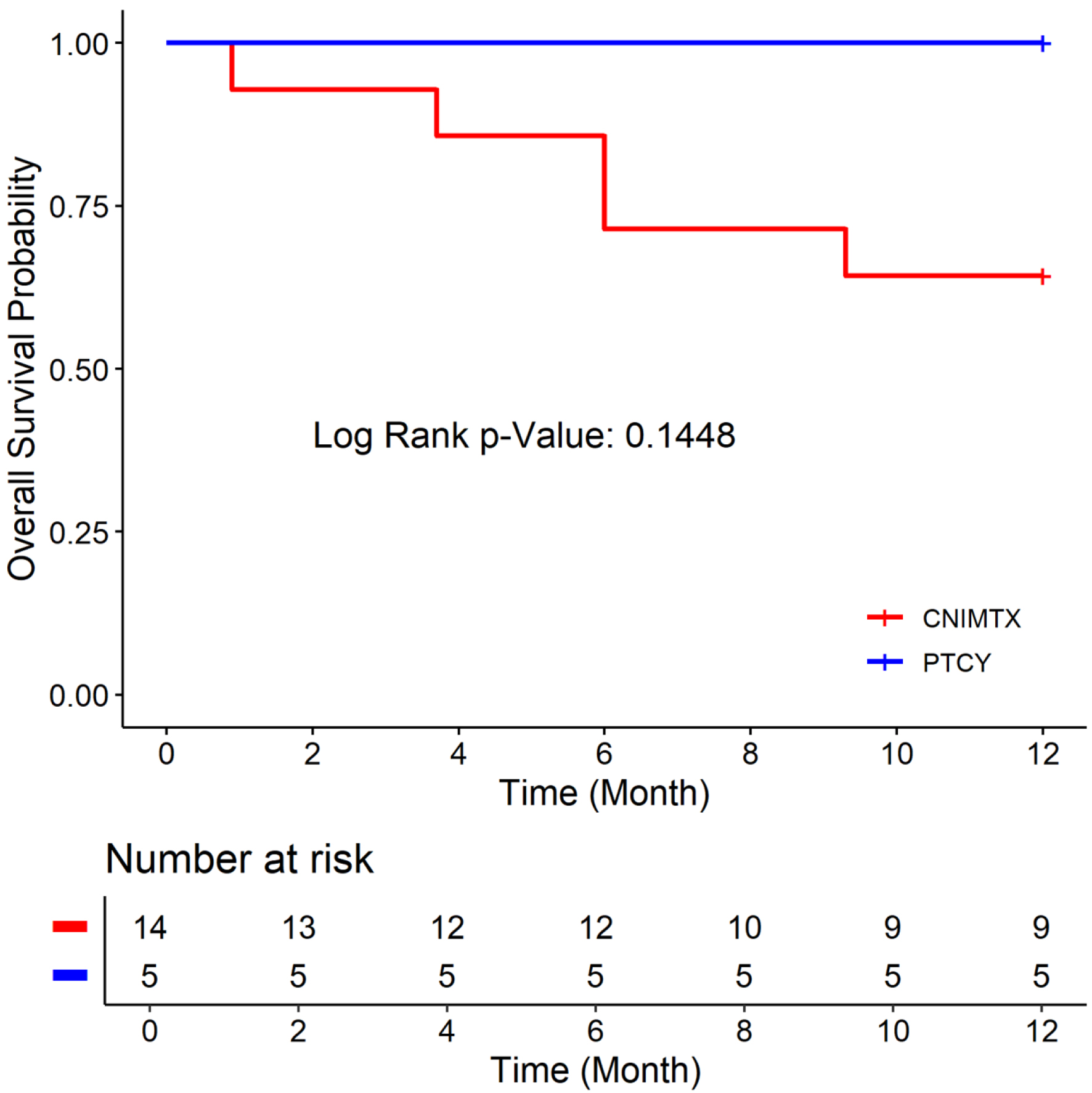
↓ Figure 1. Overall survival of the two cohorts: post-transplant cyclophosphamide (PTCY) versus calcineurin inhibitor and methotrexate (CNI-MTX).
| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Original Article
Volume 15, Number 2, April 2026, pages 71-79
Post-Transplant Cyclophosphamide Allows Allogeneic Hematopoietic Stem-Cell Transplantation Across Donor Types for Nonmalignant Hematologic Diseases
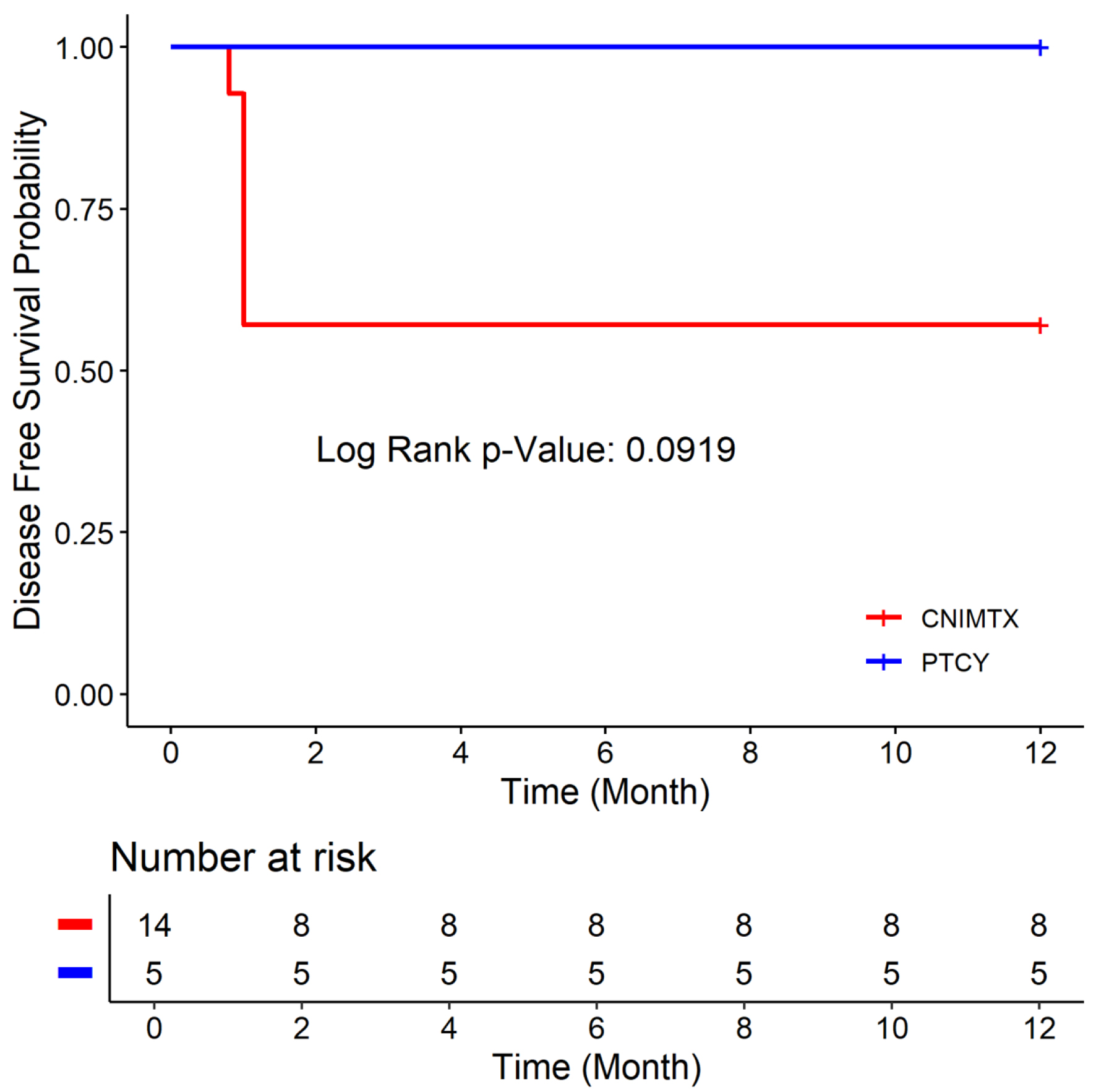
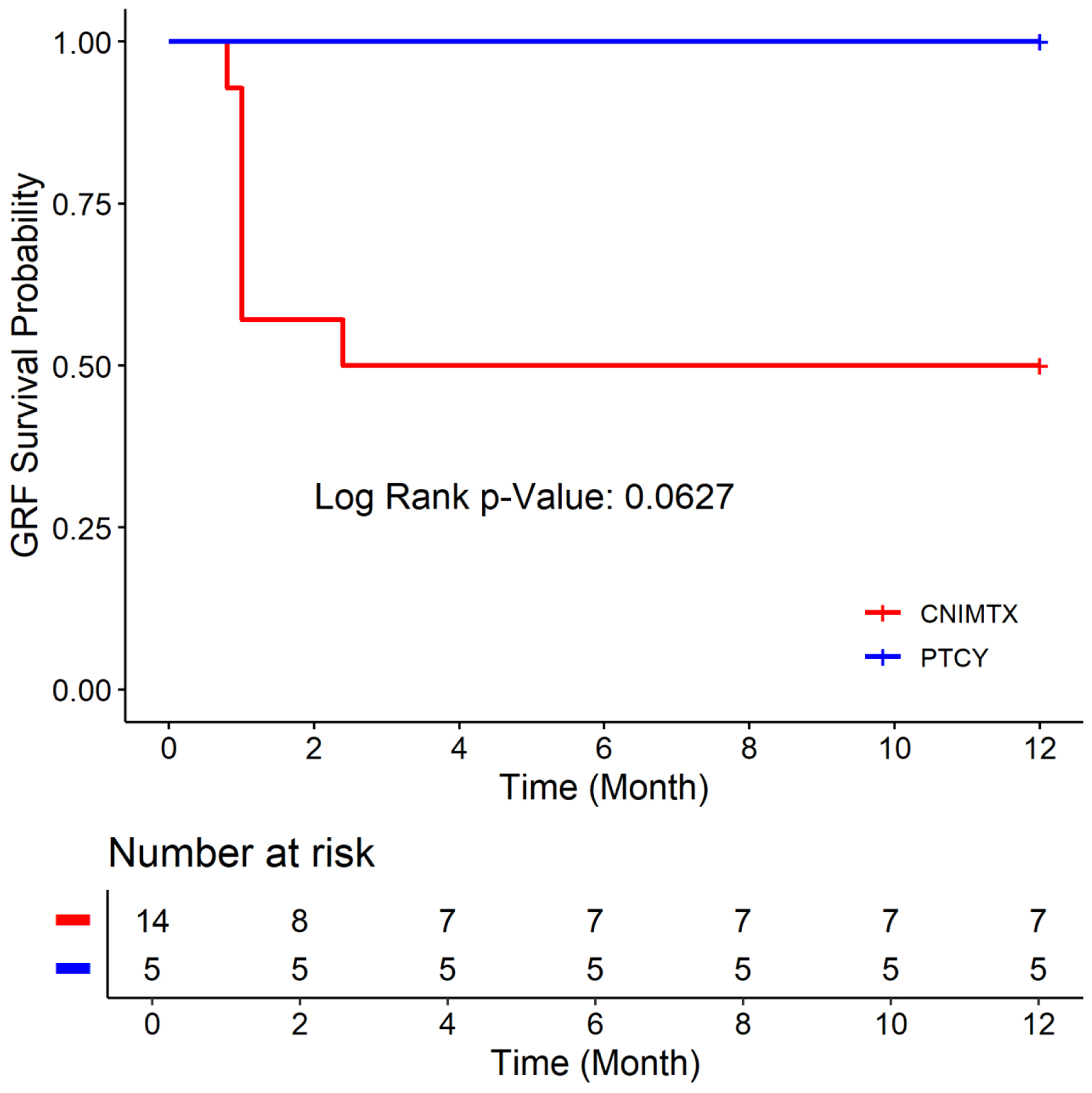
Figures

Tables
| Characteristics | PTCY (N = 5) | CNI-MTX (N = 14) | P value |
|---|---|---|---|
| ATG: rabbit anti-thymocyte globulin; CNI: calcineurin inhibitor; MTX: methotrexate; PTCY: post-transplant cyclophosphamide; TBI: total body irradiation. | |||
| Age, years, mean (minimum–maximum) | 33.8 (21–48) | 29.1 (18–58) | 0.459 |
| Gender, N (%) | 0.1409 | ||
| Female | 1 (20) | 9 (64.29) | |
| Male | 4 (80) | 5 (35.71) | |
| Diagnosis, N (%) | 0.4527 | ||
| Severe aplastic anemia | 4 (80) | 14 (100) | |
| Diamond-Blackfan anemia | 1 (20) | 0 (0) | |
| Karnofsky performance status, N (%) | 1 | ||
| 90% or more | 5 (100) | 14 (100) | |
| Donor type, N (%) | 0.1838 | ||
| Sibling donor | 1 (20) | 6 (42.86) | |
| Matched unrelated donor | 2 (40) | 7 (50) | |
| Haploidentical | 1 (20) | 0 (0) | |
| Identical twin donor (syngeneic) | 1 (20) | 0 (0) | |
| Mismatched unrelated donor (7/8) | 0 (0) | 1 (7.14) | |
| Graft source, N (%) | 0.0844 | ||
| Bone marrow | 2 (40) | 12 (85.71) | |
| Peripheral blood stem cells | 3 (60) | 2 (14.29) | |
| Chimerism, N (%) | 0.2112 | ||
| Full donor | 2 (40) | 4 (28.57) | |
| Mixed T-cell chimerism; full donor myeloid chimerism | 2 (40) | 4 (28.57) | |
| None (primary graft failure) | 0 (0) | 6 (42.86) | |
| Syngeneic donor | 1 (20) | 0 | |
| Conditioning, N (%) | 0.2723 | ||
| Cyclophosphamide and rabbit ATG | 0 (0) | 3 (21.43) | |
| Fludarabine, cyclophosphamide, rabbit ATG, TBI | 5 (100) | 11 (78.57) | |
| Age/gender/diagnosis | Prior therapy | Diagnosis to transplant (days) | CD34+/kg infused | Donor/graft source | Neutrophil engraftment (days) | Platelet engraftment (days) | Acute GVHD | Chronic GVHD | Chimerism at day 100 Myeloid/T-cell | Infection by day 100 | F/U, months |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ATG: horse anti-thymocyte globulin; BM: bone marrow; CSA: cyclosporine; DBA: Diamond-Blackfan anemia; GVHD: graft-versus-host disease; MRD: matched related donor; MUD: matched unrelated donor; PBSCs: peripheral blood stem cells; SAA: severe aplastic anemia; F/U: follow-up. | |||||||||||
| 22 years Male SAA | None | 105 | 5.96 | Syngeneic PBSC | 14 | 18 | No | No | N/A | No | 13.6 |
| 35 years Male DBA | Prednisone | 12,775 | 5.94 | MUD PBSC | 14 | 14 | No | No | 100%/100% | No | 15 |
| 48 years Male SAA | None | 118 | 5.96 | MRD PBSC | 17 | 31 | No | No | 100%/93% | No | 15.4 |
| 43 Male SAA | None | 143 | 2.16 | MUD BM | 13 | 18 | No | No | 100%/81% | No | 17.8 |
| 21 years Female SAA | ATG, CSA, Eltrombopag | 814 | 2.59 | Haploidentical related BM | 12 | 18 | No | No | 100%/100% | No | 18.7 |