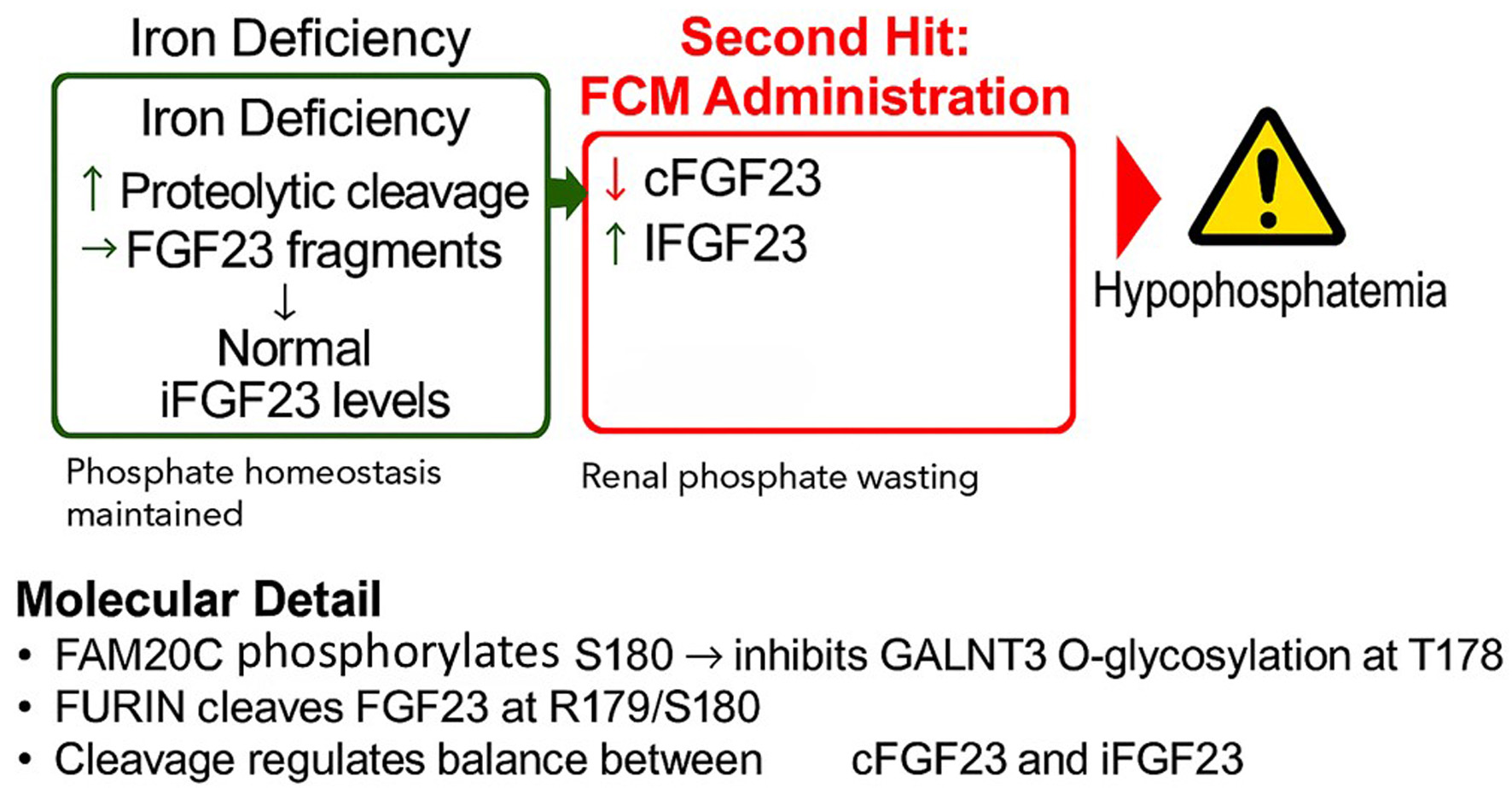
↓ Figure 1. The proposed “two-hit”
mechanism of ferric carboxymaltose (FCM)-induced hypophosphatemia. (a) In a state of iron deficiency,
transcription of the FGF23 gene is upregulated in osteocytes, leading to increased protein
production. However, this is matched by a compensatory increase in proteolytic cleavage, resulting in
high levels of inactive C-terminal FGF23 (cFGF23) fragments but normal levels of active, intact FGF23
(iFGF23). Phosphate homeostasis is maintained. (b) The administration of FCM is hypothesized to inhibit
the cleavage process. This uncouples the high production rate from degradation, leading to a surge in
circulating iFGF23, which in turn causes renal phosphate wasting and hypophosphatemia. (c) Molecular
detail of the FGF23 cleavage site. The protease FURIN cleaves the protein at R179/S180. This cleavage is
promoted by the kinase FAM20C, which phosphorylates serine 180 (S180). This phosphorylation prevents the
protective O-glycosylation of threonine 178 (T178) by the enzyme GALNT3.