| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://jh.elmerpub.com |
Case Report
Volume 000, Number 000, February 2025, pages 000-000
Donor Cell-Derived Chronic Lymphocytic Leukemia Presenting After Allogenic Hematopoietic Stem Cell Transplantation for Acute Myeloid Leukemia
Katarzyna Czempika, c , Izabela Nostera, Joanna Dziaczkowska-Suszeka, Magdalena Glowala-Kosinskab, Dariusz Kataa, Anna Kopinskaa, Grzegorz Helbiga
aDepartment of Hematology and Bone Marrow Transplantation, Medical University of Silesia, Katowice, Poland
bMaria Sklodowska-Curie National Research Institute of Oncology, Gliwice Branch, 44-102 Gliwice, Poland
cCorresponding Author: Katarzyna Czempik, Department of Hematology and Bone Marrow Transplantation, Medical University of Silesia, 40-027 Katowice, Poland
Manuscript submitted January 1, 2025, accepted February 10, 2025, published online February 21, 2025
Short title: Donor Cell-Derived CLL After HSCT
doi: https://doi.org/10.14740/jh2022
| Abstract | ▴Top |
Allogeneic hematopoietic stem cell transplantation (HSCT) is a curative approach for many hematologic disorders, and donor cell leukemia (DCL) remains a complication rarely observed after HSCT. The number of reported cases of DCL slightly exceeds 100, with acute myeloid leukemia (AML) being the most common type. To date, only a few cases of chronic lymphocytic leukemia (CLL) emerging from donor cells have been described in the literature. Here, we report two cases of CLL of donor origin, which emerged in patients after HSCT for AML. In the reported cases, patients maintained complete remission of AML after HSCT. Both donors were free of CLL before transplantation. Several years after HSCT (9 and 3 years, respectively), lymphocytosis with proven B-cell clonality was detected in recipients prompting a detailed blood analysis to be performed also in donors. CLL population was demonstrated in both cases. The first donor-recipient pair did not meet criteria for CLL treatment and eventually died from causes not related to underlying hematologic malignancy. The second couple received Bruton’s tyrosine kinase inhibitors with good disease control. The presented cases raise the question of possible clonal evolution of B-lymphocytes in donor-origin cells. Prospective screening of potential donors for pre-malignant alterations remains a matter of discussion.
Keywords: Donor cell-derived leukemia; Allogenic hematopoietic stem cell transplantation; Chronic lymphocytic leukemia
| Introduction | ▴Top |
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is a well-established curative approach for many hematological malignancies with acute myeloid leukemia (AML) being the most common indication. Post-transplant secondary malignancies are rare and include solid cancers, post-transplant lymphoproliferative diseases and donor-related leukemias. The prevalence of donor cell leukemia (DCL) is estimated to be 80 cases per 100 000 transplants [1].
Considering the lineage of DCL, myeloid malignancies are more common compared to those of lymphoid origin, and the most common type of DCLs is AML, followed by acute lymphoblastic leukemia [2].
Numerous possible models of DCL development have been proposed, involving previous presence of malignant clone in the donor or acquisition of primary or secondary mutations after the transplantation in donor-derived cells in the recipient. Postulated phenomena underlying DCL development include defective bone marrow microenvironment, impaired immune surveillance and high replicative stress. The use of growth factors within the first 100 days after transplantation, in vivo T-cell depletion and multiple allografts have been identified as significantly associated with an increased risk for DCL development [1]. Older donor age is expected to increase the risk of DCL in the recipient, with cumulative incidence of 6.3% in donors over 60 years, as clonal hematopoiesis develops with aging [3].
DCL is associated with poor survival resulting from high relapse risk with subsequent chemo-resistance [1]. The choice of treatment depends on the type of DCL. Chemotherapy remains the most common therapeutic modality, and a significant number of patients undergo second HSCT [1, 2]. More than 100 cases of DCL have been described in the literature so far [4], however fewer than 20 patients were diagnosed with chronic lymphocytic leukemia (CLL) [1, 5-10].
Herein, we present two patients who developed CLL of donor origin several years after undergoing matched sibling HSCT for AML.
| Case Reports | ▴Top |
Case 1
Investigations
A 56-year-old male was diagnosed with AML, not otherwise specified, after presenting with leukocytosis (white blood cell count: 54.2 × 109/L), moderate anemia (hemoglobin: 10.2 g/dL), and thrombocytopenia (platelet count: 65 × 109/L). His family medical history involved an undefined leukemia in his brother, who had died at the age of 18. The patient’s bone marrow cytology revealed 90% myeloblasts with typical immunophenotype (CD34-, Tdt-, CD13+dim, CD33+, CD117-, MPO+dim, CD65+dim, lysozyme+, CD14dim, CD2-, CD19-, CD22-, s+cy CD79a-). Karyotype analysis was not performed due to lack of evaluable metaphases. The biomolecular assay was positive for WT-1 oncogene and negative for the following mutations: BCR/ABL, PML/RARα, CBFβ/MYH11, RUNX1/RUNX1T1 and FLT3-ITD.
The patient received induction with DAC regimen (daunorubicin, cytarabine and cladribine), which resulted in complete remission. After consolidative treatment, he underwent HSCT from his human leukocyte antigen (HLA)-matched, 64-year-old brother. At the time of pretransplantation donor assessment, he presented with normal leukocyte count without lymphocytosis, and no abnormalities were demonstrated in biochemical tests. Moreover, his chest radiograph and abdominal ultrasound were normal. The recipient was conditioned with treosulfan with fludarabine (Treo-Flu). Graft-vs-host disease (GVHD) prophylaxis consisted of methotrexate and cyclosporine. Bone marrow aspirate at discharge showed complete remission with full donor chimerism.
Three months after HSCT, the recipient developed symptoms of acute GVHD meeting the criteria of stage I liver GVHD and stage II intestine GVHD (grade III) according to The Mount Sinai Acute GVHD International Consortium [11]. Up to 5 years after the transplantation, the recipient presented with score 1 chronic GVHD symptoms involving skin, mucosae and liver. Nine years after transplantation, an increased white blood cell count with lymphocytosis was demonstrated (10.44 × 109/L and 5.47 × 109/L, respectively). Neither lymphadenopathy nor organomegaly was found on physical examination.
Diagnosis
Flow cytometry of peripheral blood detected 60% clonal B lymphocytes. Bone marrow trephine biopsy showed 80% infiltration of CLL cells. Fluorescent in situ hybridization (FISH) on blood cells demonstrated the presence of 13q deletion and trisomy 12. The donor origin of CLL was confirmed by short tandem repeats (STR) - polymerase chain reaction (PCR) analysis of sorted CD19 bone marrow cells (Becton Dickinson FACSAria™ III sorter, purity > 98%). His donor was found to have CLL 3 months after the recipient’s diagnosis. The donor’s blood was occupied in 80% by a B-cell clonal population, presenting the same immunophenotype as the clonal population in recipient’s blood. FISH analysis of donor material has not been performed.
Treatment
Neither the recipient nor the donor met the criteria to initiate treatment for CLL.
Follow-up and outcomes
The recipient died as a result of stroke 14 years after transplantation. Unfortunately, the donor died 11 months later from an unknown reason.
Case 2
Investigations
A 59-year-old female with a medical history of hypertension was diagnosed with AML, not otherwise specified. On admission, she was leukopenic (white blood cell count: 3.37 × 109/L), with normal hemoglobin concentration and platelet count. Bone marrow aspiration showed 30% myeloblasts in the cytologic examination, with a typical immunophenotype (CD34+, HLA-DR-, Tdt-, CD13+dim, CD33+, CD117+, CD36+, MPO-, CD15-, CD65-, CD11b-, CD14-, CD64-, CD7-, CD2-, s+cy CD3-, CD56-, CD19-, CD22-, s+cy CD79a-). The molecular assay of marrow cells did not detect any of the following mutations: CBFβ/MYH11, RUNX1/RUNX1T1, FLT3-ITD and NPM1. Her karyotype was 46, XX.
She received DA regimen (daunorubicin, cytarabine) and achieved complete remission. After consolidative treatment, she underwent HSCT from her HLA-matched 50-year-old brother. His pretransplantation donor assessment was normal in terms of hematology, biochemistry or imaging studies. She was conditioned with Treo-Flu. Cyclosporine, mycophenolate mofetil and anti-thymocyte globulin were given as GVHD prophylaxis.
The recipient achieved complete remission and full donor chimerism. Her post-transplant follow-up was unremarkable except for transient symptoms of grade 1 acute GVHD (stage 1 skin GVHD) according to The Mount Sinai Acute GVHD International Consortium [11], which appeared on day 51 post-transplant. Transient Epstein-Barr virus (EBV) reactivation not requiring treatment was detected using PCR.
Three years after transplantation, leukocytosis with lymphocytosis were demonstrated (12 × 109/L and 8.42 × 109/L , respectively). Mild lymphadenopathy and hepatomegaly were present on physical examination.
Diagnosis
Flow cytometry of peripheral blood detected 85% clonal B lymphocytes. Bone marrow trephine biopsy showed 50% infiltration of CLL cells. Trisomy 12 was detected using FISH. The recipient’s immunoglobulin heavy chain variable status was unmutated. The donor origin of CLL was confirmed using STR analysis of sorted CD19 bone marrow cells (Becton Dickinson FACSAria™ III sorter, purity > 98%).
The donor was diagnosed with CLL 2 months after the recipient’s diagnosis with 84% B-cells clonality confirmed in immunophenotyping of peripheral blood. Flow cytometry revealed differences in immunophenotypic presentation of CLL in the recipient and donor (CD5+ and CD23+ in the donor’s blood immunophenotyping, CD5- and CD23- in the recipient’s blood immunophenotyping), as shown in Figure 1. FISH performed in donor material revealed trisomy 12. The donor’s immunoglobulin heavy chain variable status was unmutated.
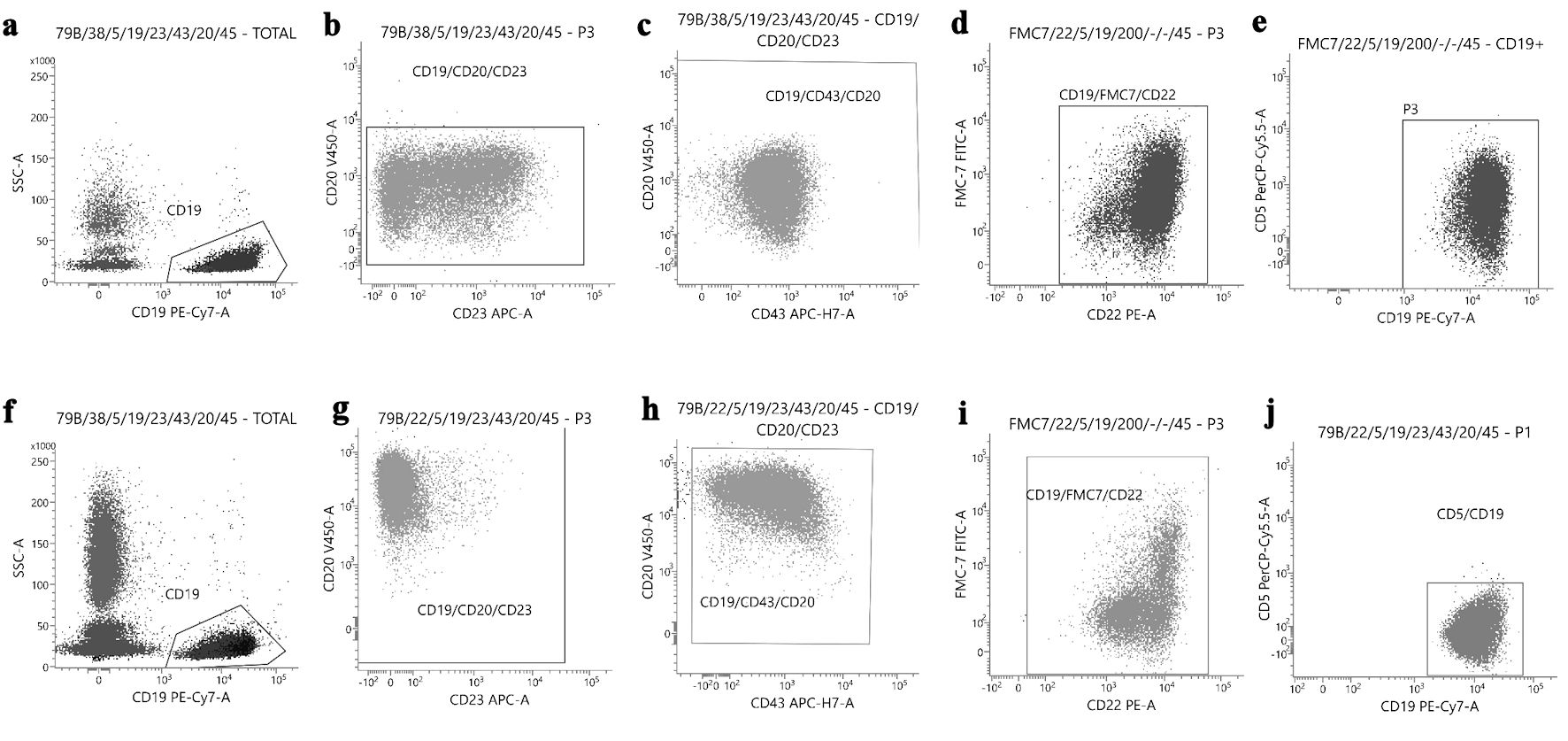 Click for large image | Figure 1. Case 2 - flow cytometry analysis of the donor (a-e) and the recipient (f-j). CD19 cells were gated on the SSC/CD19 dot plot (a, f). In blood of the donor, cells show weaker expression of CD20 (b; y-axis), but stronger expression of CD23 (b; x-axis), compared to the recipient cells (g). (c) and (h) display expression of CD43 (x-axis) versus CD20 (y-axis). CD43 expression is slightly weaker in the donor than in the recipient. (d) and (i) show a significant difference in expression of FMC-7, which is stronger in the donor (d) in comparison with the recipient (i). The expression of CD5 is present in the donor (e), contrary to the lack of CD5 expression in the recipient’s blood (j). |
Treatment
Due to the recipient’s rapidly increasing lymphocytosis, treatment with Bruton’s tyrosine kinase inhibitors was administered. Treatment has been initiated in the donor as well because of rapidly increasing lymphocytosis, progressive lymphadenopathy and general symptoms.
Follow-up and outcomes
The recipient achieved remission during her Bruton’s tyrosine kinase inhibitors treatment. Due to severe side effects, the treatment has been later discontinued, and currently the recipient remains under observation. The donor achieved remission of the disease, and the treatment with Bruton’s tyrosine kinase inhibitor is currently being continued. He also underwent basal cell carcinoma excision during his CLL treatment.
| Discussion | ▴Top |
DCL remains a rare phenomenon, first reported in 1971, yet the number of diagnosed cases has increased from 2004, which may be due to improvement of laboratory diagnostic methods and better survival rates after allo-HSCT. Donor origin of leukemia is currently confirmed mainly by STR analysis and FISH [4]. However, the prevalence of DCL may still be underestimated, especially in cases when post-transplant leukemia represents the same cell lineage as the primary disease. The total number of DCLs reported since 1971 exceeds 100 [2]. To our best knowledge, less than 20 cases of CLL emerging as DCL have been reported in the English-language medical literature to date [1, 5-10].
The proposed pathogenic mechanisms of DCL include the probable transfer of malignant clone at the time of transplantation and the infusion of normal or premalignant hemopoietic stem cells subsequently undergoing malignant transformation in the microenvironment of the recipient [4, 5, 12]. A defective hematopoietic microenvironment, being an inherent feature of the recipient or resulting from damaging effects of previous chemo- and radiotherapy, as well as impaired immune surveillance (predisposed by T-immunodeficiency following allo-HSCT, pharmacologic immunosuppression or GVHD) might play a role in leukemic transformation. No significant impact of intensity of conditioning on the incidence of DCL has been shown [1]. The proliferative demands in circumstances of rapidly repopulating entire hematopoietic system after allo-HSCT result in progressive telomere shortening and genomic instability [13]. This may act additively or synergistically with other alterations in the bone marrow environment to cause hematologic dysfunction. The role of EBV as an extrinsic factor contributing to an oncogenic transformation after allo-HSCT is confirmed in post-transplant lymphoproliferative disease, which, when associated with allo-HSCT, generally originates from donor cells [14]. However, it is not clear if EBV infection also promotes DCL development.
Clonal hematopoiesis of indeterminate potential (CHIP) is an entity defined by presence of somatic mutations in hematopoietic cells in the absence of morphologic evidence of hematologic malignancy. The prevalence of CHIP increases with age from 10.3% at the age of 60 - 64 years to 28.6% at the age of 75 - 79 years [15]. The presence of CHIP is associated with a higher incidence of hematologic malignancies. An increased donor age in allo-HSCT is associated with an elevated risk of CHIP in the recipient [12], as well as with higher incidence of DCL [16]. The sibling donors in the cases presented, particularly the donor in case 1, were of relatively advanced age. The choice of the donor in both cases was justified by faster and more convenient access to a related donor in comparison to an unrelated donor. However, it must be noted that the transplantation in case 1 was performed over a decade ago and individuals aged over 60 are not currently chosen as hematopoietic stem cells donors due to an increased risk of transplant complications.
Another disorder carrying potential risk of donor-derived CLL development is monoclonal B-cell lymphocytosis (MBL), which can putatively originate at both stem cells and more mature cells [17]. MBL is defined as clonal B-cell population in the peripheral blood with fewer than 5 × 109/L B cells and no other signs of a lymphoproliferative disorder [18]. MBL prevalence increases with age, and its frequency reaches 20% in healthy individuals older than 60 years [19]. Between 1% and 4% of individuals with CLL-type MBL per year will develop progressive disease requiring treatment [20]. MBL has been detected in patients up to 6.4 years before the development of clinically recognized leukemia [21]. A case of transmission of CLL-like MBL retrospectively detected in peripheral blood at the time of bone marrow harvest has been reported [10]. These observations indicate that testing for B-cell clonality in potential donors in the older age group may decrease the risk of transferring MBL to the recipient.
The differences in the immunophenotypic presentation of CLL in the recipient and donor in case 2 revealed in flow cytometry (Fig. 1) raise the question of possible alternative evolution of the lymphoid clone. One hypothetical explanation is the occurrence of an early lesion in a progenitor [22] or stem cell [23] in the donor before donation, and post-transplant acquisition of further mutations and alternative maturation of the clone in the donor and recipient [24, 25]. In another scenario, a mature B-cell clone could be transplanted, with subsequent transformation in the recipient, which could be indicated by the putative loss of CD5 and CD23 expression in the recipient’s B cells, which is a common event in CLL transformation to diffuse large B-cell lymphoma [25]. Since immunophenotyping was not performed in the donor’s material at the time of donation, the presence of monoclonal lymphocytosis at this time cannot be ruled out.
Eleven cases of donor cell-derived CLL reported in the literature so far are summarized in Table 1 [1, 5-10]. Donor follow-up data were available in nine cases, and in all of them, as well as in two of our cases, the leukemic clone was found in the donor after allo-HSCT. Engel et al obtained detailed follow-up data for 25 donors, of whom five cases were diagnosed with the same hematologic malignancy as their recipients (CLL in four cases, chronic myeloid leukemia in one case) [1]. In contrast, numerous cases of other DCLs, predominantly AML, have been reported, but hematological malignancies in donors have been excluded [1, 5], which apparently has never been reported in donor-recipient pairs where the recipient developed CLL in cells of donor origin. Such consistency of results in CLL patients and their donors may suggest the presence of preleukemic alterations in donors’ cells at donation. Unfortunately, flow cytometry studies were not performed at donation.
 Click to view | Table 1. Review of the Literature (Reported Cases of Donor-Derived CLL) |
The phenomenon of leukemia arising from donor cells after allo-HSCT raises the question of donor stem cells screening before transplantation in searching of clonal hematopoiesis. Some authors postulate screening for CHIP in allogenic donors older than age 50 years to minimize the risk of DCL [26], whereas others conclude that the current level of evidence is insufficient to implement prospective screening of donors for CHIP in clinical practice [27, 28]. Further investigation is needed to assess the clinical impact of CHIP.
Learning points
DCL is a diagnostic challenge, and it may be misdiagnosed as a relapse of primary leukemia when presenting the same lineage. The pathogenesis of DCL remains incompletely understood, yet it constitutes an interesting model for studies on leukemogenesis. Prospective screening of donors for CHIP and MBL remains a matter of discussion.
Acknowledgments
Special acknowledgement to all the healthcare and medical laboratory professionals involved in the diagnosis and care of the patients described in this report.
Financial Disclosure
None to declare
Conflict of Interest
None to declare
Informed Consent
The recipient’s informed consent in case 1 and case 2, and donor’s consent in case 2 for publication of the case reports were obtained. The donor in case 1 is deceased. Since obtaining consent prior to his death was not possible, all data have been fully anonymized to ensure the privacy, and identifiable information was not included in this report.
Author Contributions
Katarzyna Czempik: data acquisition, literature review, writing of the manuscript. Izabela Noster: patient care and clinical case management. Joanna Dziaczkowska-Suszek: patient’s diagnosis, interpretation of the results, preparation of figure, editorial assistance. Magdalena Glowala-Kosinska: patient’s diagnosis. Dariusz Kata: conceptualization of the study, patient care, editorial assistance. Anna Kopinska: patient care, clinical case management, editorial assistance. Grzegorz Helbig: supervision of the project, editorial assistance, critical review of the manuscript.
Data Availability
The data supporting the findings of this study are available within the article.
Abbreviations
HSCT: hematopoietic stem cell transplantation; DCL: donor cell leukemia; AML: acute myeloid leukemia; CLL: chronic lymphocytic leukemia; GVHD: graft-vs-host disease; EBV: Epstein-Barr virus; FISH: fluorescent in situ hybridization; STR: short tandem repeats; PCR: polymerase chain reaction; CHIP: clonal hematopoiesis of indeterminate potential; MBL: monoclonal B-cell lymphocytosis; ALL: acute lymphoblastic leukemia; BM: bone marrow; CML: chronic myelocytic leukemia; HLA: human leukocyte antigen; HSC: hematopoietic stem cells; IgH: immunoglobulin heavy chain; IgK: immunoglobulin kappa light chain; MDS: myelodysplastic syndrome; ND: no data; NHL: non-Hodgkin lymphoma; PB: peripheral blood; Sib: sibling donor; VNTR: variable number of tandem repeats
| References | ▴Top |
- Engel N, Rovo A, Badoglio M, Labopin M, Basak GW, Beguin Y, Guyotat D, et al. European experience and risk factor analysis of donor cell-derived leukaemias/MDS following haematopoietic cell transplantation. Leukemia. 2019;33(2):508-517.
doi pubmed - Suarez-Gonzalez J, Martinez-Laperche C, Kwon M, Balsalobre P, Carbonell D, Chicano M, Rodriguez-Macias G, et al. Donor cell-derived hematologic neoplasms after hematopoietic stem cell transplantation: a systematic review. Biol Blood Marrow Transplant. 2018;24(7):1505-1513.
doi pubmed - Gondek LP, Zheng G, Ghiaur G, DeZern AE, Matsui W, Yegnasubramanian S, Lin MT, et al. Donor cell leukemia arising from clonal hematopoiesis after bone marrow transplantation. Leukemia. 2016;30(9):1916-1920.
doi pubmed - Zhang S, Li L, Cao W, Li Y, Jiang Z, Yu J, Wan D. Donor cell leukemia/myelodysplastic syndrome after allogeneic stem cell transplantation: a rare phenomenon with more challenges for hematologists. Hematology. 2021;26(1):648-651.
doi pubmed - Sala-Torra O, Hanna C, Loken MR, Flowers ME, Maris M, Ladne PA, Mason JR, et al. Evidence of donor-derived hematologic malignancies after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2006;12(5):511-517.
doi pubmed - Pavletic SZ, Zhou G, Sobocinski K, Marti G, Doney K, DiPersio J, Feremans W, et al. Genetically identical twin transplantation for chronic lymphocytic leukemia. Leukemia. 2007;21(12):2452-2455.
doi pubmed - Nahi H, Jansson M, Sander B, Ljungman P, Hagglund H. Transmission of chronic lymphocytic leukaemia from a blood stem cell sibling donor to the recipient. Br J Haematol. 2008;143(5):751-753.
doi pubmed - Perz JB, Ritgen M, Moos M, Ho AD, Kneba M, Dreger P. Occurrence of donor-derived CLL 8 years after sibling donor SCT for CML. Bone Marrow Transplant. 2008;42(10):687-688.
doi pubmed - Flandrin-Gresta P, Callanan M, Nadal N, Jaubert J, Cornillon J, Guyotat D, Campos L. Transmission of leukemic donor cells by allogeneic stem cell transplantation in a context of familial CLL: should we screen donors for MBL? Blood. 2010;116(23):5077-5078.
doi pubmed - Ferrand C, Garnache-Ottou F, Collonge-Rame MA, Larosa F, Blanc M, Behar C, Giannoli C, et al. Systematic donor blood qualification by flow cytometry would have been able to avoid CLL-type MBL transmission after unrelated hematopoietic stem cell transplantation. Eur J Haematol. 2012;88(3):269-272.
doi pubmed - Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, Martin P, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005;11(12):945-956.
doi pubmed - Taniguchi R, Muramatsu H, Okuno Y, Suzuki K, Obu S, Nakatochi M, Shimamura T, et al. Comprehensive genetic analysis of donor cell derived leukemia with KMT2A rearrangement. Pediatr Blood Cancer. 2018;65(2):e26823.
doi pubmed - Boettcher S, Wilk CM, Singer J, Beier F, Burcklen E, Beisel C, Ventura Ferreira MS, et al. Clonal hematopoiesis in donors and long-term survivors of related allogeneic hematopoietic stem cell transplantation. Blood. 2020;135(18):1548-1559.
doi pubmed - Wiseman DH. Donor cell leukemia: a review. Biol Blood Marrow Transplant. 2011;17(6):771-789.
doi pubmed - Frick M, Chan W, Arends CM, Hablesreiter R, Halik A, Heuser M, Michonneau D, et al. Role of donor clonal hematopoiesis in allogeneic hematopoietic stem-cell transplantation. J Clin Oncol. 2019;37(5):375-385.
doi pubmed - Kato M, Yamashita T, Suzuki R, Matsumoto K, Nishimori H, Takahashi S, Iwato K, et al. Donor cell-derived hematological malignancy: a survey by the Japan Society for Hematopoietic Cell Transplantation. Leukemia. 2016;30(8):1742-1745.
doi pubmed - von Beck K, von Beck T, Ferrell PB, Jr., Bick AG, Kishtagari A. Lymphoid clonal hematopoiesis: implications for malignancy, immunity, and treatment. Blood Cancer J. 2023;13(1):5.
doi pubmed - Strati P, Shanafelt TD. Monoclonal B-cell lymphocytosis and early-stage chronic lymphocytic leukemia: diagnosis, natural history, and risk stratification. Blood. 2015;126(4):454-462.
doi pubmed - Nieto WG, Almeida J, Romero A, Teodosio C, Lopez A, Henriques AF, Sanchez ML, et al. Increased frequency (12%) of circulating chronic lymphocytic leukemia-like B-cell clones in healthy subjects using a highly sensitive multicolor flow cytometry approach. Blood. 2009;114(1):33-37.
doi pubmed - Rawstron AC. Monoclonal B-cell lymphocytosis. Hematology Am Soc Hematol Educ Program. 2009;2009:430-439.
doi pubmed - Landgren O, Albitar M, Ma W, Abbasi F, Hayes RB, Ghia P, Marti GE, et al. B-cell clones as early markers for chronic lymphocytic leukemia. N Engl J Med. 2009;360(7):659-667.
doi pubmed - Damm F, Mylonas E, Cosson A, Yoshida K, Della Valle V, Mouly E, Diop M, et al. Acquired initiating mutations in early hematopoietic cells of CLL patients. Cancer Discov. 2014;4(9):1088-1101.
doi pubmed - Kikushige Y, Ishikawa F, Miyamoto T, Shima T, Urata S, Yoshimoto G, Mori Y, et al. Self-renewing hematopoietic stem cell is the primary target in pathogenesis of human chronic lymphocytic leukemia. Cancer Cell. 2011;20(2):246-259.
doi pubmed - Chiorazzi N, Ferrarini M. Cellular origin(s) of chronic lymphocytic leukemia: cautionary notes and additional considerations and possibilities. Blood. 2011;117(6):1781-1791.
doi pubmed - Milpied P, Nadel B, Roulland S. Premalignant cell dynamics in indolent B-cell malignancies. Curr Opin Hematol. 2015;22(4):388-396.
doi pubmed - DeZern AE, Gondek LP. Stem cell donors should be screened for CHIP. Blood Adv. 2020;4(4):784-788.
doi pubmed - Gibson CJ, Lindsley RC. Stem cell donors should not be screened for clonal hematopoiesis. Blood Adv. 2020;4(4):789-792.
doi pubmed - Wilk CM, Manz MG, Boettcher S. Clonal hematopoiesis in hematopoietic stem cell transplantation. Curr Opin Hematol. 2021;28(2):94-100.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.